| Content: | 50 assays / 100 assays |
| Shipping Temperature: | Ambient |
| Storage Temperature: |
Short term stability: 2-8oC, Long term stability: See individual component labels |
| Stability: | > 2 years under recommended storage conditions |
| Analyte: | Total Starch |
| Assay Format: | Spectrophotometer |
| Detection Method: | Absorbance |
| Wavelength (nm): | 510 |
| Signal Response: | Increase |
| Linear Range: | 4 to 100 μg of D-glucose per assay |
| Limit of Detection: | 0.18 g/100 g total starch “as is” |
| Total Assay Time: | ~ 90 min |
| Application examples: | Cereal flours, food products and other materials. |
| Method recognition: | AACC Method 76-13.01, AOAC Method 996.11, ICC Standard Method No. 168 and RACI Standard Method |
The K-TSTA-50A pack size has been discontinued (read more)
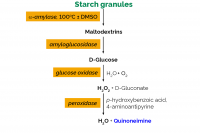
The Total Starch (AA/AMG) Assay Kit is used for the determination of total starch in cereal flours and food products. AOAC Method 996.11, AACC Method 76-13.01.
This kit now contains an improved α-amylase that allows the amylase incubations to be performed at pH 5.0 (as well as pH 7.0).
See our full range of dietary fiber and starch assay kits.

- Very competitive price (cost per test)
- All reagents stable for > 2 years after preparation
- Rapid reaction
- Mega-Calc™ software tool is available from our website for hassle-free raw data processing
- Standard included
Measurement of available carbohydrates, digestible, and resistant starch in food ingredients and products.
McCleary, B. V., McLoughlin, C., Charmier, L. M. J. & McGeough, P. (2019). Cereal Chemistry, 97(1), 114-137.
Background and objectives: The importance of selectively measuring available and unavailable carbohydrates in the human diet has been recognized for over 100 years. The levels of available carbohydrates in diets can be directly linked to major diseases of the Western world, namely Type II diabetes and obesity. Methodology for measurement of total carbohydrates by difference was introduced in the 1880s, and this forms the basis of carbohydrate determination in the United States. In the United Kingdom, a method to directly measure available carbohydrates was introduced in the 1920s to assist diabetic patients with food selection. The aim of the current work was to develop simple, specific, and reliable methods for available carbohydrates and digestible starch (and resistant starch). The major component of available carbohydrates in most foods is digestible starch. Findings: Simple methods for the measurement of rapidly digested starch, slowly digested starch, total digestible starch, resistant starch, and available carbohydrates have been developed, and the digestibility of phosphate cross‐linked starch has been studied in detail. The resistant starch procedure developed is an update of current procedures and incorporates incubation conditions with pancreatic α‐amylase (PAA) and amyloglucosidase (AMG) that parallel those used AOAC Method 2017.16 for total dietary fiber. Available carbohydrates are measured as glucose, fructose, and galactose, following complete and selective hydrolysis of digestible starch, maltodextrins, maltose, sucrose, and lactose to glucose, fructose, and galactose. Sucrose is hydrolyzed with a specific sucrase enzyme that has no action on fructo‐oligosaccharides (FOS). Conclusions: The currently described “available carbohydrates” method together with the total dietary fiber method (AOAC Method 2017.16) allows the measurement of all carbohydrates in food products, including digestible starch. Significance and novelty: This paper describes a simple and specific method for measurement of available carbohydrates in cereal, food, and feed products. This is the first method that provides the correct measurement of digestible starch and sucrose in the presence of FOS. Such methodology is essential for accurate labeling of food products, allowing consumers to make informed decisions in food selection.
Hide AbstractMeasurement of Starch: Critical evaluation of current methodology.
McCleary, B. V., Charmier, L. M. J. & McKie, V. A. (2018). Starch‐Stärke, 71(1-2), 1800146.
Most commonly used methods for the measurement of starch in food, feeds and ingredients employ the combined action of α‐amylase and amyloglucosidase to hydrolyse the starch to glucose, followed by glucose determination with a glucose oxidase/peroxidase reagent. Recently, a number of questions have been raised concerning possible complications in starch analytical methods. In this paper, each of these concerns, including starch hydrolysis, isomerisation of maltose to maltulose, effective hydrolysis of maltodextrins by amyloglucosidase, enzyme purity and hydrolysis of sucrose and β‐glucans have been studied in detailed. Results obtained for a range of starch containing samples using AOAC Methods 996.11 and 2014 .10 are compared and a new simpler format for starch measurement is introduced. With this method that employs a thermostable α-amylase (as distinct from a heat stable α-amylase) which is both stable and active at 100°C and pH 5.0, 10 samples can be analysed within 2 h, as compared to the 6 h required with AOAC Method 2014.10.
Hide AbstractMcCleary, B. V., Charnock, S. J., Rossiter, P. C., O’Shea, M. F., Power, A. M. & Lloyd, R. M. (2006). Journal of the Science of Food and Agriculture, 86(11), 1648-1661.
Procedures for the measurement of starch, starch damage (gelatinised starch), resistant starch and the amylose/amylopectin content of starch, β-glucan, fructan, glucomannan and galactosyl-sucrose oligosaccharides (raffinose, stachyose and verbascose) in plant material, animal feeds and foods are described. Most of these methods have been successfully subjected to interlaboratory evaluation. All methods are based on the use of enzymes either purified by conventional chromatography or produced using molecular biology techniques. Such methods allow specific, accurate and reliable quantification of a particular component. Problems in calculating the actual weight of galactosyl-sucrose oligosaccharides in test samples are discussed in detail.
Hide AbstractMcCleary, B. V., Gibson, T. S. & Mugford, D. C. (1997). Journal of AOAC International, 80, 571-579.
An American Association of Cereal Chemists/AOAC collaborative study was conducted to evaluate the accuracy and reliability of an enzyme assay kit procedure for measurement of total starch in a range of cereal grains and products. The flour sample is incubated at 95 degrees C with thermostable alpha-amylase to catalyze the hydrolysis of starch to maltodextrins, the pH of the slurry is adjusted, and the slurry is treated with a highly purified amyloglucosidase to quantitatively hydrolyze the dextrins to glucose. Glucose is measured with glucose oxidase-peroxidase reagent. Thirty-two collaborators were sent 16 homogeneous test samples as 8 blind duplicates. These samples included chicken feed pellets, white bread, green peas, high-amylose maize starch, white wheat flour, wheat starch, oat bran, and spaghetti. All samples were analyzed by the standard procedure as detailed above; 4 samples (high-amylose maize starch and wheat starch) were also analyzed by a method that requires the samples to be cooked first in dimethyl sulfoxide (DMSO). Relative standard deviations for repeatability (RSD(r)) ranged from 2.1 to 3.9%, and relative standard deviations for reproducibility (RSD(R)) ranged from 2.9 to 5.7%. The RSD(R) value for high amylose maize starch analyzed by the standard (non-DMSO) procedure was 5.7%; the value was reduced to 2.9% when the DMSO procedure was used, and the determined starch values increased from 86.9 to 97.2%.
Hide AbstractCollaborative evaluation of a simplified assay for total starch in cereal products (AACC Method 76-13).
McCleary, B. V., Gibson, T. S. & Mugford, D. C. (1997). Cereal Foods World, 42, 476-480.
A procedure for the quantitative analysis of total starch in plant materials has been developed and subjected to a comprehensive interlaboratory study involving 32 laboratories, in accordance with the protocol for collaborative studies recommended by American Association of Cereal Chemists and AOAC International. The method involved treatment of a sample at approximately 95°C with thermostable α-amylase to obtain starch depolymerization and solubilisation. The slurry is then treated with purified amyloglucosidase to give quantitative hydrolysis of the starch fragments to glucose, which is measured with glucose oxidase/peroxidase reagent. Test samples used in the interlaboratory study included modified and native starches, cereal flours and brans, processed cereal products, animal feeds, and plant material. Results were statistically analysed according to AOAC International guidelines (1). The procedure was shown to be highly repeatable (relative standard deviation 2.1-3.9%) and reproducible (relative standard deviation 2.9-5.0%), and on the basis of these results has gained first approval status with AACC (AACC Method 76-13) and approval as AOAC Method 986.11. The method is more robust than a method previously reported (AACC Method 76-12), and 20 samples can be analysed within 2 hr.
Hide AbstractMcCleary, B. V., Gibson, T. S., Solah, V. & Mugford, D. C. (1994). Cereal Chemistry, 71(5), 501-505.
The precision of an enzymatic procedure for analysis of total starch in cereal flours and products was determined in a comprehensive inter-laboratory study involving 29 laboratories. Test samples represented a range of sample types, including modified and native starches, cereal flours and brans, processed cereal products, animal feeds, and plant material. Results were statistically analyzed according to AOAC guidelines. The procedure was shown to be highly repeatable (relative standard deviation 1.5-7.3%) and reproducible (relative standard deviation 4.1-11.3%). It is now available, in a slightly modified form, as an assay kit. The assay, therefore, provides a convenient alternative to existing procedures for quantitative measurement of starch in cereal products.
Hide AbstractQuantitative measurement of total starch in cereal flours and products.
McCleary, B. V., Solah, V. & Gibson, T. S. (1994). Journal of Cereal Science, 20(1), 51-58.
A rapid and quantitative method has been developed for the determination of total starch in a wide range of materials, including high-amylose maize starches and food materials containing resistant starch. The method allows the analysis of 20 samples in 3 h. A single assay can be performed in 2 h. For a range of samples, the total starch values obtained with this method were significantly higher than those obtained with current standard methods. Two assay formats have been developed. In assay format 1, the sample is incubated solubilised with the chaotropic agent dimethyl sulphoxide (DMSO) to gelatinise the starch, which is then solubilised and partially depolymerised by controlled incubation at ∼ 100°C with a defined level of thermostable alpha-amylase. This allowed near-complete solubilisation of most starches. The remaining starch is then solubilised and the starch fragments are converted to maltose and maltotriose by the combined action of highly purified pullulanase and beta-amylase. After volume adjustment and filtration (if necessary), the maltooligo-saccharides are hydrolysed by high-purity amyloglucosidase to glucose, which is measured with a glucose oxidase/peroxidase reagent. This assay format gave quantitative starch determination in all native starch samples, including high-amylose maize starches. In assay format 2, which is applicable to most starches and cereal flours, the DMSO pre-treatment step is omitted. Samples containing glucose and/or maltosaccharide are pre-washed with aqueous ethanol before analysis.
Hide AbstractSpray-drying of chickpea aquafaba with biopolymers: Improving physical and techno-functional properties for meringue production.
da Silva-Padilha, M. P., Semedo, J. M., da Silva Toledo, C. C., da Silva, V. M. & Hubinger, M. D. (2025). International journal of biological macromolecules, 319, 145426.
Chickpea aquafaba can replace eggs, but its industrial handling is challenging. Drying results in highly hygroscopic powders, affecting product quality during storage. To address this, the addition of biopolymeric carriers to aquafaba during spray drying may reduce water adsorption in the product. This study aimed to produce spray-dried chickpea aquafaba using carriers such as maltodextrin (MD), gum Arabic (GA), and their combination (1:1, MD:GA), followed by the characterization of the powders' physical and techno-functional properties. Among the formulations, the use of MD significantly reduced the hygroscopicity of the powder. Powders produced with the MD:GA mixture showed a critical moisture content of 0.042 g water/g dry solids, compared to 0.087 g water/g dry solids for the control, according to the GAB model, indicating good physical stability. The addition of GA improved the emulsifying capacity of the aquafaba powders while reducing their foaming and gelling capacities. In contrast, MD enhanced foaming capacity but did not improve emulsification. As proof of concept, selected powders were incorporated into egg-free meringue formulations, with rheological properties modulated by the addition of low concentration of biopolymeric carriers. Overall, the use of carriers during spray drying chickpea aquafaba improved the physical properties of powders and modulated their techno-functional properties.
Hide AbstractDeproteinization and controlled starch prehydrolysis in intact pulse cotyledon cells regulates starch retrogradation and digestion.
Mao, M., Martinez, M. M., Tuncil, Y., Ding, L., Chen, C., Fu, X., Huang, Q. & Zhang, B. (2025). Carbohydrate Polymers, 367, 123922.
Intact pulse cotyledon cells are known for their intact cellular structure and dense protein matrix, leading to slower starch digestibility. However, the retrogradation and digestibility of retrograded intracellular starch has rarely been investigated, and our preliminary results indicated the difficulty of intracellular starch retrogradation. To test mechanistic principles of intracellular starch retrogradation, intact pulse cells were extracted and treated with protease followed by pancreatic α-amylase (0-20 min) and pullulanase (24 h) prehydrolysis, and then controlled retrogradation (4°C, 5 d). Protein hydrolysis had little effect on starch retrogradation and digestibility, showing no change in the crystallinity, retrogradation enthalpy change and extent of digestion, compared to the cooked sample. Meanwhile, additional 5 min of α-amylase hydrolysis maximized the retrogradation of intracellular starch likely by producing perfect chain length for retrogradation, which then exhibited the lowest digestion rate and extent (0.0172 min-1 and 29.8%). Further increasing α-amylase hydrolysis reduced crystallinity and enthalpy, leading to decreased retrogradation and increased digestibility. These findings shed new light on the mechanism underlying intracellular starch retrogradation and the extending application of retrograded starch with low propensity to negatively affect texture.
Hide AbstractEvaluating the Impact of Flour Protein Content on the Quality of Wholegrain Refrigerated Dough.
Malalgoda, M., Stefanson, R., Swaminathan, I., Valsalan, A., Malunga, L. N. & Garcia, I. G. (2025). Cereal Chemistry., 102(4), 756-765.
Background and Objectives: Refrigerated dough products offer convenience, but are often produced using refined flour. However, wholewheat flour-based products provide health benefits by increasing dietary fiber intake. This study aims to determine the optimal flour protein content for developing clean label refrigerated dough using wholegrain Canada Western Red Spring (CWRS) wheat. Three CWRS wheat samples with varying protein contents: Sample 1–Average Protein (S1-AP) (13.30%), Sample 2—Intermediate High Protein (S2-IHP) (14.91%), and Sample 3—High Protein (S3-HP) (16.48%), were analyzed. The proximate composition of wholegrain flour samples was determined using standard methods. Dough mixing and starch pasting properties were evaluated using micro-dough LAB and RVA, respectively. Dough samples were prepared and stored at 4°C for 35 days, with subsets baked and assessed for end-use quality every 7 days. Xylanase activity and dough syruping were monitored throughout storage. Findings: S3-HP exhibited reduced starch pasting properties, shorter dough development time, and lower stability. S2-IHP demonstrated better mixing stability and lower mixing tolerance index, indicating superior dough handling properties. Xylanase activity was highest in S1-AP at 35 days and lowest in S2-IHP. Dough syruping increased significantly during the first 14 days and stabilized thereafter. Bread quality declined with storage, with volume decreasing and firmness increasing, especially in S1-AP. Lightness (L*) decreased over time in all samples. Conclusions: S2-IHP, which showed intermediate/moderately high protein content, was identified as the most suitable for wholegrain refrigerated dough, as it maintained superior end-use quality characteristics compared to the other samples.
Hide AbstractImpact of extrusion-induced protein molecular rearrangement on cooking qualities, in vitro digestibility and gluten allergenicity of durum wheat pasta.
Jia, B., Devkota, L., Sissons, M., Attenborough, E., Ni, D. & Dhital, S. (2025). Food Chemistry, 492, 145293.
The cooking quality and digestibility of pasta are influenced by its protein structures. However, how shear forces affect protein molecular structure leading to pasta quality and the allergenicity of pasta is not well understood. This study explored the impact of varying screw speeds (150 rpm and 550 rpm), which directly influence specific mechanical energy (SME) input during extrusion, on protein molecular weight distribution, cooking properties, and starch and protein digestibility in both regular durum wheat pasta and gluten-enriched pasta. Pasta produced at 550 rpm exhibited reduced water absorption and cooking time but significantly increased cooking loss, accompanied by a decrease in disulphide bonds. Size-exclusion chromatography (SEC) and sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) results indicated that high screw speed caused the insolubility of glutenins and globulins while increasing the solubility of gliadins. Additionally, these structural changes corresponded to decreased ionic and hydrogen bonding and increased hydrophobic interactions. Although starch and protein enzymic digestibility was decreased with added gluten, the increased screw speed enhanced their digestion. Proteomic mapping by liquid chromatography-tandem mass spectrometry (LC-MS/MS analysis) revealed a reduction in the number of identified proteins and allergens in the digestate of pasta produced at 550 rpm, while the number of identified peptides increased. Additionally, added gluten delayed protein hydrolysis during the gastric phase and offset differences due to screw speed. This understanding aids in optimizing processing and formulations, balancing efficiency with pasta's nutritional and cooking quality.
Hide AbstractSustainable 2, 3-Butanediol Production from Brewer’s Spent Grains via Bacterial Co-culture and Fermentation Strategy Optimization.
Rama, E., López-Linares, J. C., García-Cubero, M. T., Coca, M., Dragone, G. & Mussatto, S. I. (2025). Environmental Technology & Innovation, 39, 104339.
The use of lignocellulosic biomass in fermentation processes is often hindered by inhibitory compounds generated during the biomass pretreatment, which negatively impact microbial performance. This study aimed to develop a sustainable bioprocess for producing 2,3-butanediol (2,3-BDO) from brewer’s spent grains (BSG) using the Generally Recognized as Safe (GRAS) bacterium Paenibacillus polymyxa. Monoculture of P. polymyxa and co-culture fermentation using P. polymyxa and Pseudomonas putida were explored, with co-culture being proposed as a sustainable detoxification alternative to conventional chemical methods. In addition, to evaluate the impact of process configuration on 2,3-BDO production, three saccharification and fermentation approaches were compared: separate hydrolysis and fermentation (SHF), simultaneous saccharification and fermentation (SSF), and simultaneous saccharification and co-fermentation (SSCF). Among these, the SSCF approach combined with co-culture fermentation demonstrated the highest performance, achieving a 2,3-BDO production of 20.94 g/L. These results underscore the potential of microbial co-cultivation as an effective strategy to overcome the inhibitory effects of lignocellulosic hydrolysates, offering a promising route for sustainable 2,3-BDO production from BSG.
Hide AbstractExtensive remodeling during Chlamydomonas reinhardtii zygote maturation leads to highly resistant zygospores.
Cardador, M., Krüger, S., Dunker, S., Brakel, A., Hoffmann, R., Nagel, R., Jakob, T., Goss, R. & Sasso, S. (2025). The Plant Journal, 121(3), e17238.
The unicellular soil alga Chlamydomonas reinhardtii forms diploid zygotes during its sexual cycle. The process of a zygote maturing into a highly resistant zygospore remains poorly understood despite its importance for survival under adverse environmental conditions. Here we describe the detailed timeline of morphological and physiological changes during zygote maturation in darkness on ammonium-free Tris-acetate-phosphate agar plates. The formation of a multilayered cell wall is primarily responsible for the increase in cell size in the first few days after zygote formation. Desiccation and freezing tolerance also develop in the period 3-7 days. Photosynthetic and respiratory activity decrease to reach minimal levels after 7-10 days, accompanied by a partial dedifferentiation of the chloroplast that includes chlorophyll degradation followed by the possible disappearance of the pyrenoid. In contrast to the decreasing concentrations of most carotenoids in the first few days after zygote formation, ketocarotenoids can first be detected after 3 days and their accumulation is completed after 10 days. Furthermore, the zygote degrades a large proportion of its starch and enriches oligosaccharides that may serve as osmoprotectants. The storage lipid triacylglycerol is accumulated at the expense of thylakoid membrane lipids, which mirrors the conversion of a metabolically active cell into a dormant spore on the metabolic level. Taken together, zygote maturation is a multifaceted process that yields mature zygospores after ~ 3 weeks. This work sheds light on the complete time course of the remodeling of a photosynthetically active eukaryotic cell into a dormant, highly resistant spore.
Hide AbstractOptimization of a craft ale-type beer enriched with cañihua malt (Chenopodium pallidicaule) and banana passionfruit juice (Passiflora tripartita var. mollisima).
Paucar-Menacho, L. M., Salvador-Reyes, R., Castillo-Martinez, W. E., Lavado-Cruz, A., Verona-Ruiz, A., Campos-Rodriguez, J., Acosta-Coral, K., Simpalo-Lopez, W. D., López-Rodriguez, W. & Quezada-Berrú, S. (2025). Heliyon, 11(4), e42610.
The global expansion of the craft beer market has driven the incorporation of native ingredients to enhance the sensory and nutritional profiles of beer. This study focused on optimizing a craft Ale-type beer enriched with Cañihua Malt (CM) and Banana Passionfruit Juice (BPJ) using a D-optimal experimental design. The aim was to evaluate how varying concentrations of these ingredients (CM: 15-25 %, BPJ: 5-15 %) influence the physicochemical, technological, and sensory attributes of beer. Results demonstrated that the malting process significantly improved the nutritional composition of cañihua, increasing fiber content (23.32 g/100 g), phenolic compounds (141.13 mg GAE/100 g), GABA (229.48 mg/100 g), and antioxidant capacity (1975.41 μmol TE/g dw). These enhancements positively affected the physicochemical properties of beer, especially foam stability and body. The addition of BPJ significantly modified the physicochemical characteristics of beer, particularly by reducing the pH and increasing the acidity. Sensory analysis showed high consumer acceptance, with positive evaluations for aroma, appearance, and body, particularly in samples containing moderate levels of CM (15-16%) and BPJ (5-10%). Optimization using the desirability function identified ideal concentrations of 24%-25% CM and 5% BPJ, achieving a balance in critical parameters such as foam stability, density, pH, and bitterness. These findings underscore the potential to combine CM and BPJ to develop a distinctive craft beer with enhanced sensory attributes and nutritional benefits.
Hide AbstractGermination time: impact on nutritional, thermal, and paste properties of red rice.
Heberle, T., do Nascimento, L. Á., Wang, Y. J., Colussi, R., de Souza, J. F., Fajardo, A. R. & Mesko, M. F. (2025). Journal of the Science of Food and Agriculture, 105(9), 4941-4948.
Background: The study investigates how varying germination times affect the nutritional, thermal, and pasting properties of red rice. Germination was conducted over 32 h, with aliquots collected every 2 h starting from 18 h for property evaluation. Results: At all evaluated germination times, the red rice samples exhibited low to intermediate amylose levels, while resistant starch content remained relatively constant (approximately 15-20%). Structural changes in the starch were observed: the peak viscosity decreased, maximum gelatinization of the granules occurred at 76°C, and spectroscopic bands characteristic of this polysaccharide were detected at 3400 cm−1 (O-H stretching) and within the 1150-1000 cm−1 range (C-OH and C-O-C stretching). Protein and fiber content remained unchanged throughout the process, whereas carbohydrate content varied by 10 g kg−1. Protein digestibility increased after 26 h and continued to rise until the end of germination, while starch digestibility decreased throughout the process, particularly between 24 and 26 h. Conculsion: The thermal properties of the grains remained unchanged, as did the starch digestibility, suggesting the potential application of this flour in gluten-free bakery products.
Hide Abstract