| Content: | 2,000 Units |
| Shipping Temperature: | Ambient |
| Storage Temperature: | 2-8oC |
| Formulation: | In 3.2 M ammonium sulphate |
| Physical Form: | Suspension |
| Stability: | > 1 year under recommended storage conditions |
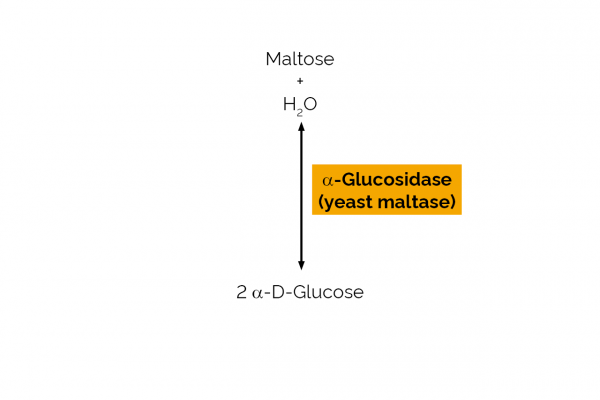
| Enzyme Activity: | α-Glucosidase |
| EC Number: | 3.2.1.20 |
| CAZy Family: | GH13 |
| CAS Number: | 9001-42-7 |
| Synonyms: | alpha-glucosidase; alpha-D-glucoside glucohydrolase |
| Source: | Yeast |
| Molecular Weight: | 52,000 |
| Concentration: | Supplied at ~ 1,000 U/mL |
| Expression: | From Yeast |
| Specificity: | Hydrolysis of terminal, non-reducing (1,4)-linked α-D-glucose residues with release of D-glucose. |
| Specific Activity: | ~ 120 U/mg (40oC, pH 6.8 on pNP-α-Glucosidase) |
| Unit Definition: | One Unit of α-glucosidase activity is defined as the amount of enzyme required to produce one µmole of p-nitrophenol from pNP-α-Glucosidase (10 mM) in sodium phosphate buffer (100 mM), pH 6.8 at 40oC. |
| Temperature Optima: | 40oC |
| pH Optima: | 6.8 |
| Application examples: | Applications in carbohydrate research and in the food and feeds, brewing and biofuels industries. |
High purity α-Glucosidase (yeast maltase) for use in research, biochemical enzyme assays and analytical testing applications.
Show all of our CAZyme products.
Diastatic power and maltose value: a method for the measurement of amylolytic enzymes in malt.
Charmier, L. M., McLoughlin, C. & McCleary, B. V. (2021). Journal of the Institute of Brewing, In Press.
A simple method for measurement of the amylolytic activity of malt has been developed and fully evaluated. The method, termed the Maltose Value (MV) is an extension of previously reported work. Here, the MV method has been studied in detail and all aspects of the assay (sample grinding and extraction, starch hydrolysis, maltose hydrolysis and determination as glucose) have been optimised. The method is highly correlated with other dextrinising power methods. The MV method involves extraction of malt in 0.5% sodium chloride at 30°C for 20 minutes followed by filtration; incubation of an aliquot of the undiluted filtrate with starch solution (pH 4.6) at 30°C for 15 min; termination of reaction with sodium hydroxide solution; dilution of sample in an appropriate buffer; hydrolysis of maltose with a specific α-glucosidase; glucose determination and activity calculation. Unlike all subsequent reducing sugar methods, the maltose value method measures a defined reaction product, maltose, with no requirement to use equations to relate analytical values back to Lintner units. The maltose value method is the first viable method in 130 years that could effectively replace the 1886 Lintner method.
Hide AbstractDevelopment and validation of two analytical strategies for the determination of glucosides of acidic herbicides in cereals and oilseed matrices.
Aloisi, I. & Mol, H. (2023). Analytical and Bioanalytical Chemistry, 1-11.
The aim of the present research was the development and validation of a selective and reliable method for the indirect and direct determination of acidic herbicide glucosides. Enzymatic deconjugation was investigated as a mild alternative to harsh alkaline hydrolysis. Various enzymatic options for deconjugation were exploited. One out of nine tested specific enzymes proved to be practical and repeatable for different matrices and concentration ranges, leading to the complete deconjugation of the glucosides. The method was validated according to the SANTE/11312/2021 guideline for cereals and oilseeds and for a rice-based infant formula. Additionally, for four acidic herbicide glucosides available on the market, a quantitative method for direct determination of the intact glucosides was optimized and validated. In both methods, the average recoveries were within 70–120%. The limits of quantification (LOQ) achieved were 10 µg kg−1 and 2.5 µg kg−1 for the intact glucosides and the free acids in cereal and oilseeds. For the rice-based infant formula, the LOQ was 1 µg kg−1 (3 µg kg−1 for dichlorprop). To confirm its applicability, the deconjugation approach was tested for fifteen samples (cereals, oilseeds, and citrus) with incurred residues. Comparisons were made between the method without deconjugation, and two methods with deconjugation, the here proposed enzymatic deconjugation and the more commonly used alkaline hydrolysis. The inclusion of enzymatic deconjugation during sample preparation led to an increase up to 2.7-fold compared to analysis without deconjugation. Enzymatic deconjugation resulted in comparable results to alkaline hydrolysis for 13 out of 15 samples.
Hide AbstractPreliminary Phytochemical Screening, In Vitro Antidiabetic, Antioxidant Activities, and Toxicity of Leaf Extracts of Psychotria malayana Jack.
Nipun, T. S., Khatib, A., Ahmed, Q. U., Nasir, M. H. M., Supandi, F., Taher, M. & Saiman, M. Z. (2021). Plants, 10(12), 2688.
Psychotria malayana Jack belongs to the Rubiacea and is widespread in Southeast Asian countries. It is traditionally used to treat diabetes. Despite its potential medicinal use, scientific proof of this pharmacological action and the toxic effect of this plant are still lacking. Hence, this study aimed to investigate the in vitro antidiabetic and antioxidant activities, toxicity, and preliminary phytochemical screening of P. malayana leaf extracts by gas chromatography-mass spectrometry (GC-MS) after derivatization. The antidiabetic activities of different extracts of this plant were investigated through alpha-glucosidase inhibitory (AGI) and 2-NBDG glucose uptake using 3T3-L1 cell line assays, while the antioxidant activity was evaluated using DPPH and FRAP assays. Its toxicological effect was investigated using the zebrafish embryo/larvae (Danio rerio) model. The mortality, hatchability, tail-detachment, yolk size, eye size, beat per minute (BPM), and body length were taken into account to observe the teratogenicity in all zebrafish embryos exposed to methanol extract. The LC50 was determined using probit analysis. The methanol extract showed the AGI activity (IC50 = 2.71 ± 0.11 μg/mL), insulin-sensitizing activity (at a concentration of 5 µg/mL), and potent antioxidant activities (IC50 = 10.85 μg/mL and 72.53 mg AAE/g for DPPH and FRAP activity, respectively). Similarly, the water extract exhibited AGI activity (IC50 = 6.75 μg/mL), insulin-sensitizing activity at the concentration of 10 μg/mL, and antioxidant activities (IC50 = 27.12 and 33.71 μg/mL for DPPH and FRAP activity, respectively). The methanol and water extracts exhibited the LC50 value higher than their therapeutic concentration, i.e., 37.50 and 252.45 µg/mL, respectively. These results indicate that both water and methanol extracts are safe and potentially an antidiabetic agent, but the former is preferable since its therapeutic index (LC50/therapeutic concentration) is much higher than for methanol extracts. Analysis using GC-MS on derivatized methanol and water extracts of P. malayana leaves detected partial information on some constituents including palmitic acid, 1,3,5-benzenetriol, 1-monopalmitin, beta-tocopherol, 24-epicampesterol, alpha-tocopherol, and stigmast-5-ene, that could be a potential target to further investigate the antidiabetic properties of the plant. Nevertheless, isolation and identification of the bioactive compounds are required to confirm their antidiabetic activity and toxicity.
Hide AbstractDiurnal patterns of growth and transient reserves of sink and source tissues are affected by cold nights in barley.
Barros, K. A., Esteves‐Ferreira, A. A., Inaba, M., Meally, H., Finnan, J., Barth, S., Davis, S. J. & Sulpice, R. (2020). Plant, Cell & Environment, 43(6), 1404-1420.
Barley is described to mostly use sucrose for night carbon requirements. To understand how the transient carbon is accumulated and utilized in response to cold, barley plants were grown in a combination of cold days and/or nights. Both daytime and night cold reduced growth. Sucrose was the main carbohydrate supplying growth at night, representing 50–60% of the carbon consumed. Under warm days and nights, starch was the second contributor with 26% and malate the third with 15%. Under cold nights, the contribution of starch was severely reduced, due to an inhibition of its synthesis, including under warm days, and malate was the second contributor to C requirements with 24-28% of the total amount of carbon consumed. We propose that malate plays a critical role as an alternative carbon source to sucrose and starch in barley. Hexoses, malate, and sucrose mobilization and starch accumulation were affected in barley elf3 clock mutants, suggesting a clock regulation of their metabolism, without affecting growth and photosynthesis however. Altogether, our data suggest that the mobilization of sucrose and malate and/or barley growth machinery are sensitive to cold.
Hide AbstractIdentification of α-glucosidase inhibitors from Clinacanthus nutans leaf extract using liquid chromatography-mass spectrometry-based metabolomics and protein-ligand interaction with molecular docking.
Murugesu, S., Ibrahim, Z., Ahmed, Q. U., Uzir, B. F., Yusoff, N. I. N., Perumal, V., Saari, K., Khatib, A. & Khatib, A. (2019). Journal of Pharmaceutical Analysis, 9(2), 91-99.
The present study used in vitro and in silico techniques, as well as the metabolomics approach to characterise α-glucosidase inhibitors from different fractions of Clinacanthus nutans. C. nutans is a medicinal plant belonging to the Acanthaceae family, and is traditionally used to treat diabetes in Malaysia. n-Hexane, n-hexane: ethyl acetate (1:1, v/v), ethyl acetate, ethyl acetate: methanol (1:1, v/v), and methanol fractions were obtained via partitioning of the 80% methanolic crude extract. The in vitro α-glucosidase inhibitory activity was analyzed using all the fractions collected, followed by profiling of the metabolites using liquid chromatography combined with mass spectrometry. The partial least square (PLS) statistical model was developed using the SIMCA P+14.0 software and the following four inhibitors were obtained: (1) 4,6,8-Megastigmatrien-3-one; (2) N-Isobutyl-2-nonen-6,8-diynamide; (3) 1′,2′-bis(acetyloxy)-3′,4′-didehydro-2′-hydro-β, ψ-carotene; and (4) 22-acetate-3-hydroxy-21-(6-methyl-2,4-octadienoate)-olean-12-en-28-oic acid. The in silico study performed via molecular docking with the crystal structure of yeast isomaltase (PDB code: 3A4A) involved a hydrogen bond and some hydrophobic interactions between the inhibitors and protein. The residues that interacted include ASN259, HID295, LYS156, ARG335, and GLY209 with a hydrogen bond, while TRP15, TYR158, VAL232, HIE280, ALA292, PRO312, LEU313, VAL313, PHE314, ARG315, TYR316, VAL319, and TRP343 with other forms of bonding.
Hide AbstractRapid investigation of α-glucosidase inhibitory activity of Clinacanthus nutans leaf using infrared fingerprinting.
Murugesu, S., Ahmed, Q. U., Uzir, B. F., Yusoff, N. I. N., Perumal, V., Ibrahim, Z., Abas, F., Saari, K. & Khatib, A. (2019). Vibrational Spectroscopy, 100, 22-29.
The analytical method used in the quality control of Clinacanthus nutans leaves has not been well developed. Therefore, this study aimed to develop a simple analytical method to predict α-glucosidase inhibitory activity of this herb based on its infrared fingerprinting. The dried extracts obtained from maceration using solvents with different polarities were evaluated for the α-glucosidase inhibitory activity and analysed through infrared spectroscopy. Multivariate data analysis was performed by correlating the bioactivity and infrared spectrum of each extract using partial least square method. The loading plot from multivariate data analysis revealed that C-H and C=O infrared signals from terpenoids in the extract were positively correlated with the α-glucosidase inhibitory activity. The developed partial least square model was validated through a testing on the external samples. The result concludes that the developed model is valid and capable of predicting α-glucosidase inhibitory activity of the external samples.
Hide AbstractRevanna, R., Turnbull, M. H., Shaw, M. L., Wright, K. M., Butler, R. C., Jameson, P. E. & McCallum, J. A. (2013). Journal of the Science of Food and Agriculture, 93(10), 2470-2477.
Background: Non-structural carbohydrate (NSC; glucose, fructose, sucrose and fructan) composition of onions (Allium cepa L.) varies widely and is a key determinant of market usage. To analyse the physiology and genetics of onion carbohydrate metabolism and to enable selective breeding, an inexpensive, reliable and practicable sugar assay is required to phenotype large numbers of samples. Results: A rapid, reliable and cost-effective microplate-based assay was developed for NSC analysis in onions and used to characterise variation in tissue hexose, sucrose and fructan content in open-pollinated breeding populations and in mapping populations developed from a wide onion cross. Sucrose measured in microplates employing maltase as a hydrolytic enzyme was in agreement with HPLC-PAD results. The method revealed significant variation in bulb fructan content within open-pollinated ‘Pukekohe Longkeeper’ breeding populations over a threefold range. Very wide segregation from 80 to 600 g kg−1 in fructan content was observed in bulbs of F2 genetic mapping populations from the wide onion cross ‘Nasik Red × CUDH2150’. Conclusion: The microplate enzymatic assay is a reliable and practicable method for onion sugar analysis for genetics, breeding and food technology. Open-pollinated onion populations may harbour extensive within-population variability in carbohydrate content, which may be quantified and exploited using this method. The phenotypic data obtained from genetic mapping populations show that the method is well suited to detailed genetic and physiological analysis.
Hide Abstract