| Content: | 100 assays per kit |
| Shipping Temperature: | Ambient |
| Storage Temperature: |
Short term stability: 2-8oC, Long term stability: See individual component labels |
| Stability: | > 2 years under recommended storage conditions |
| Analyte: | β-Amylase |
| Assay Format: | Spectrophotometer |
| Detection Method: | Absorbance |
| Wavelength (nm): | 400 |
| Signal Response: | Increase |
| Limit of Detection: | 0.05 U/mL |
| Reproducibility (%): | ~ 3% |
| Reaction Time (min): | ~ 10 min |
| Application examples: | Cereal flours, malts and other materials. |
| Method recognition: | Modification of RACI Standard Method |
The Betamyl-3; β-Amylase test kit is suitable for the specific measurement and analysis of β-amylase in malt flour.
See more of our amylase assay kits and other kit products for measurement of enzyme activities.

- Very cost effective
- All reagents stable for > 2 years as supplied
- Only enzymatic kit available
- Very specific
- Simple format
- Rapid reaction
- Mega-Calc™ software tool is available from our website for hassle-free raw data processing
- Standard included
A novel enzymatic method discriminating wheat pre-harvest sprouting from Late Maturity alpha-amylase.
Mangan, D., Draga, A., Ivory, R., Cornaggia, C., Blundell, M., Howitt, C., McCleary, B. V. & Ral, J. P. (2022). Journal of Cereal Science, 105, 103480.
The primary quality assessor of wheat grain is the Hagberg Falling Number (FN) method. This is a viscometric test surrogate for α-amylase activity. Despite being used for over sixty years, FN has been increasingly scrutinised due to its low throughput, poor reproducibility and inability to differentiate between the causes of low FN including Pre-harvest Sprouting (PHS) and Late Maturity α-Amylase (LMA). Our study describes initial efforts to analyse a specific wheat flour set tailored for the identification of enzymatic candidates that would allow discrimination between PHS and LMA affected grains. Using the sensitive enzyme-coupled assay substrate R-AMGR3, results suggest that α-glucosidase (exo-α-glucosidase) is a potential enzyme marker candidate to specifically detect sprouted but not LMA-affected grain.
Hide AbstractDiastatic power and maltose value: a method for the measurement of amylolytic enzymes in malt.
Charmier, L. M., McLoughlin, C. & McCleary, B. V. (2021). Journal of the Institute of Brewing, In Press.
A simple method for measurement of the amylolytic activity of malt has been developed and fully evaluated. The method, termed the Maltose Value (MV) is an extension of previously reported work. Here, the MV method has been studied in detail and all aspects of the assay (sample grinding and extraction, starch hydrolysis, maltose hydrolysis and determination as glucose) have been optimised. The method is highly correlated with other dextrinising power methods. The MV method involves extraction of malt in 0.5% sodium chloride at 30°C for 20 minutes followed by filtration; incubation of an aliquot of the undiluted filtrate with starch solution (pH 4.6) at 30°C for 15 min; termination of reaction with sodium hydroxide solution; dilution of sample in an appropriate buffer; hydrolysis of maltose with a specific α-glucosidase; glucose determination and activity calculation. Unlike all subsequent reducing sugar methods, the maltose value method measures a defined reaction product, maltose, with no requirement to use equations to relate analytical values back to Lintner units. The maltose value method is the first viable method in 130 years that could effectively replace the 1886 Lintner method.
Hide AbstractPrediction of potential malt extract and beer filterability using conventional and novel malt assays.
Cornaggia, C., Evans, D. E., Draga, A., Mangan, D. & McCleary, B. V. (2019). Journal of Institute of Brewing, 125(3), 294-309.
Colourimetric assays were used to measure the activities of six key hydrolases endogenous to barley: β‐glucanase, xylanase, cellulase, α-amylase, beta‐amylase and limit dextrinase. The analysed barley malt samples were previously characterised by 27 conventional malt quality descriptors. Correlations between enzymatic activities and brewing parameters such as extract yield, fermentability, viscosity and filterability were investigated. A single extraction protocol for all six hydrolases was optimised and used for multi‐enzyme analysis using fully automatable assay formats. A regression analysis between malt parameters was undertaken to produce a relationship matrix linking enzyme activities and conventional malt quality descriptors. This regression analysis was used to inform a multi‐linear regression approach to create predictive models for extract yield, apparent attenuation limit, viscosity and filterability using the Small‐scale Wort rapId Filtration Test (SWIFT) and two different mashing protocols – Congress and a modified infusion mash at 65oC (MIM 65oC). It was observed that malt enzyme activities displayed significant correlations with the analysed brewing parameters. Both starch hydrolases and cell wall hydrolase activities together with modification parameters (i.e. Kolbach index) were found to be highly correlated with extract yield and apparent attenuation limit. Interestingly, it was observed that xylanase activity in malts was an important predictor for wort viscosity and filterability. It is envisaged that the automatable measurement of enzyme activity could find use in plant breeding progeny selection and for routine assessment of the functional brewing performance of malt batches. This analytical approach would also contribute to brewing process consistency, product quality and reduced processing times.
Hide AbstractMcCleary, B. V. & Codd, R. (1989). Journal of Cereal Science, 9(1), 17-33.
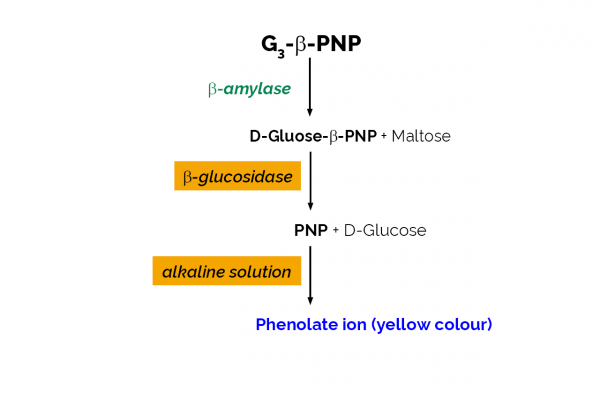
A procedure previously developed for the assay of cereal-flour β-amylase has been improved and standardised. The improved procedure uses the substrate p-nitrophenyl maltopentaose (PNPG5) in the presence of near saturating levels of α-glucosidase. PNPG5 is rapidly hydrolysed by β-amylase but less readily by cereal α-amylases. The substrate is hydrolysed by β-amylase to maltose and p-nitrophenyl maltotriose (PNPG3). With the levels of α-glucosidase used in the substrate mixture, PNPG3 is rapidly cleaved to glucose and p-nitrophenol, whereas PNPG5 is resistant to hydrolysis by the α-glucosidase. The assay procedure has been standardised for several β-amylases and the exact degree of interference by cereal α-amylases determined. The procedure can be readily applied to the selective measurement of β-amylase activity in cereal and malted cereal-flours.
Hide AbstractDiastatic Activity of German Hop Cultivars with Respect to Variety, Crop Year, and Separated Hop Cone Parts.
Wietstock, P. C., Michalek, D., Treetzen, T., Pinto, M. B. C., Biendl, M. & Gibson, B. (2025). ACS Food Science & Technology, 5(6), 2408-2416.
Dry hopping of beer can result in unintended refermentation, also known as hop creep, because of intrinsic hop diastatic activity. The objective of the work described herein was to determine the enzymatic activity across 16 different hop cultivars grown in Germany in crop years 2019, 2020, and 2021. Optimized enzyme kit protocols were used to quantitate hop α-, β-amylase, amyloglucosidase, and limit dextrinase activities, while a recently published method measured hop diastatic activity. Clear varietal distinctions exist, and hops of harvests 2019, 2020, and 2021 were subsequently classified into three groups depending on their enzymatic activity. With respect to different harvest years, the results imply an annual influence on the amylolytic activity of hops in principle, but more monitoring is needed. Processing methods such as pelletization and storage under different conditions showed a minimal impact on enzymatic activity. Based on further sampling from hops of the harvest 2022, it was observed that differences among hop fractions are pronounced, with the vegetative material and strig exhibiting higher enzymatic activity compared to the lupulin fraction.
Hide AbstractMalting and brewing process optimization of elite lines of triticale for beer production.
Nocente, F., De Francesco, G., Marconi, O., Floridi, S., Latini, A., Cantale, C., Galeffi, P., Ammar, K. & Gazza, L. (2024). Food and Bioprocess Technology, 1-10.
Triticale stands out as a valuable food ingredient due to its nutritional and functional attributes derived from both wheat and rye. The growing demand for agrifood diversity has resulted in increased interest in triticale for food and beverage production, and as research and development of new breeding lines continue, triticale is likely to play a more prominent role in the food industry, contributing to healthy, diversified, and sustainable food systems. In this context, triticale is suitable for beer production, which is traditionally made from barley malt, but can be produced by the addition of alternative grains. In this study, five triticale lines were tested both as unmalted, to produce an auto-saccharified wort, and as malted grain. Upon the malting process, line 7 was found to be the most effective in terms of malting performance; three beer formulations with different percentages of addition (i.e., 40, 70, and 100%) of malted selected triticale lines were produced in an experimental pilot plant and characterized for percentage of alcohol, foam stability, haze, and visual and taste profile. The beer containing 40% malted triticale was judged to be particularly interesting in terms of its stable foam, lighter color, greater attenuation, and lower turbidity than those produced from 70% or 100% malted triticale. This formulation could have potential for scale-up in industrial production and for the market.
Hide AbstractEnhancing malting performance of harder barley varieties through ultrasound treatment.
Qin, Q., Zhang, L., Yin, H., Yu, J., Hu, S., Zhang, Z. & Liu, J. (2024). Ultrasonics Sonochemistry, 105, 106860.
Harder kernels of barley are regarded as one of the factors that restrict water and enzyme movement within the endosperm during malting. A comprehensive study of two domestic varieties was performed for evaluating malting quality. Both β-glucan and total protein content of the Chinese domestic barley (Ganpi-6 and Kenpi-14) were significantly higher than Copeland. Grain hardness of the Chinese domestic barley was higher and water uptake ratio was lower compared with the Copeland. During germination, the expression levels of NCED1, NCED2 (major key regulatory enzymes for abscisic acid biosynthesis genes) were higher, whereas gibberelic acid (GA) synthesis genes (GA20ox1, GA2ox3, GA3ox2) were lower in the Ganpi-6, Kenpi-14 compared with Copeland. These two domestic barley varieties also showed significantly lower limit dextrinase and β-glucanase activity compared with Copeland. Ultrasound treatment improved the malting quality of Ganpi-6 by enhancing water uptake and GA synthesis gene expression increased. Therefore, these findings provided insights into the future direction on the utilization of ultrasonication for the applications towards the improvement of the harder barley variety.
Hide AbstractSynthesis in situ of heteropolysaccharide by Levilactobacillus brevis AM7 during fermentation of oat and hemp and its effect on the techno-functional properties of oat yogurt type model.
Wang, Y., Mehmood, S., Maina, N. H., Katina, K. & Coda, R. (2024). Food Hydrocolloids, 147, 109416.
Plant raw materials, such as oat bran concentrate (OBC), oat flour (OF), and hemp protein concentrate (HP), are significant sources of dietary fibers and proteins with high nutritional quality. Exopolysaccharides (EPS) synthesized by lactic acid bacteria (LAB) during fermentation are natural texturing agents that can replace hydrocolloid additives (e.g., carrageenan, guar gum, and pectin). This study aimed to evaluate EPS synthesis in OBC, OF, and HP during fermentation with Levilactobacillus brevis AM7 supplemented with glucose. Monosaccharide analysis identified that EPS from L. brevis AM7 was a heteropolysaccharide (HePS) consisting of rhamnose, mannose, glucose, and glucose-amine. Rheological analysis revealed that HePS production led to increased viscosity and viscoelastic moduli (G′ and G″) of the fermented materials compared to control. Fermented OBC exhibited the most stable structure among the three materials. Fermented OBC also showed the lowest acidity level, whereas fermented HP exhibited intensive acidification. This was likely due to the more abundant flour endogenous sugars (sucrose, raffinose, and stachyose) in HP and their fast utilization during fermentation. A yogurt alternative product was prepared from fermented OBC milk, showing a high LAB cell density, a pH of 4.2, and a significantly higher viscosity than the chemically acidified control. The in situ synthesized HePS counteracted the viscosity loss and inhibited syneresis of the yogurt alternative product during four days of storage at 4°C. Overall, the use of L. brevis AM7 producing HePS enabled improved techno-functional properties of plant raw materials and showed potential for application in yogurt alternatives.
Hide AbstractInfluences of Lactiplantibacillus plantarum and Saccharomyces cerevisiae fermentation on the nutritional components, flavor property and lipid-lowering effect of highland barley.
Bai, J., He, L., Zhang, J., Gu, X., Wu, B., Wang, A., Zhu, Y., Zhang, J., Zhao, Y. & Xiao, X. (2024). Journal of Future Foods, 4(3), 258-266.
Highland barley is a well-known cereal in Qinghai-Tibet Plateau area with high nutritional value, which has been reported to be a health-promoting grain for the obesity and the diabetes. Fermentation by certain microbiota can improve the flavor property and nutritional characteristics. In the present study, Lactiplantibacillus plantarum and Saccharomyces cerevisiae were singly or jointly applied to ferment highland barley, and the profile of volatile substances and lipid-lowering effects of the respective extracts were analyzed. Results indicated that either L. plantarum or S. cerevisiae or co-fermentation could consume the polysaccharides of highland barley to provide energy, and dramatically increase the contents of total protein and polyphenol. Gas chromatography-mass spectrometry (GC-MS) analysis revealed that the presence of S. cerevisiae was critical for production of the pleasant flavors, especially for the ethyl ester substances including hexadecanoic acid ethyl, hexanoic acid ethyl ester and so on. Meanwhile, we found that fermented highland barley extracts by L. plantarum exhibited stronger lipid-lowering effects in Caenorhabditis elegans than that by S. cerevisiae, while the co-fermentation not only emitted pleasant odors but also exerted high hypolipidemic function. In all, co-fermentation by L. plantarum and S. cerevisiae was proposed to be a promising processing to improve the flavor and functional properties of highland barley.
Hide AbstractComparative analysis of malt quality and starch characteristics of three South Korean barley cultivars.
Park, J., Chung, H. J., Park, H. Y., Park, H. J. & Oh, S. K. (2023). Food Science and Biotechnology, 1-11.
In this study, malt was produced in pilot-scale facilities and conditioned using three barley (Hordeum vulgare L.) cultivars in South Korea (Heugho, Hopum, and Kwangmaeg). Quality and starch characteristics were compared. The starch content was considerably reduced in all malts. Coleoptile elongation was higher in Heugho (HHM; 85.7% ± 12.6%) and Hopum (HPM; 83.9% ± 10.7%) than in Kwangmaeg (KMM; 78.1% ± 9.9%) malt. Malt yield ranged from 81.8 to 84.9%, with no significant difference. All samples presented type A crystallinity, and granules showed discoid shapes. After malting, the mono- and di-saccharide contents (not including sucrose) were increased. The fermentable sugar level was the highest in HHM, whereas non-fermentable sugar was the highest in KMM. These results suggest that HPM enables efficient scarification based on the rapid degradation of starch, while Heugho barley and HHM have a high potential for beer and malt production, respectively.
Hide AbstractVegan shrimp alternative made with pink oyster and lion’s mane mushrooms: Nutritional profiles, presence of conjugated phenolic acids, and prototyping.
Meyer, F., Hutmacher, A., Lu, B., Steiger, N., Nyström, L. & Narciso, J. O. (2023). Current Research in Food Science, 7, 100572.
The increasing demand for seafood is responsible for many environmental impacts, especially caused by aquaculture. Shrimp accounts for a substantial part of seafood production and therefore also for negative effects associated with it. This work aimed to develop a mushroom-based shrimp analogue with a texture similar to shrimp using the fruiting bodies of pink oyster mushroom (Pleurotus djamor) and lion’s mane (Hericium erinaceus). Three flushes of pink oyster mushrooms and a first flush of lion’s mane mushroom were analysed regarding their nutritional composition and whether they are suitable shrimp alternatives. The two mushrooms are rich in proteins (~32% and ~26% w/w for the first flush of pink oyster and lion’s mane, respectively). The protein content of pink oyster mushroom decreased and the dietary fibre content increased across the different flushes. The antioxidants in the mushrooms were extracted using different methods, whereby aqueous extracts mostly excelled in terms of antioxidant activity. Hydrolysis confirmed the presence of conjugated p-coumaric acid in both mushrooms and possibly conjugated caffeic acid in pink oyster. Texture analysis results of the prototypes were close to the values of fried shrimp. However, although the sensory qualities of the final prototypes were perceived as similar to shrimp, further improvements in the recipe are necessary to make the prototypes indistinguishable from shrimp.
Hide AbstractEffects by the Hard Water Boiling of Chalky Rice in Terms of Texture Improvement and Ca Fortification.
Nakamura, S. & Ohtsubo, K. I. (2023). Foods, 12(13), 12132510.
In the present paper, we investigated the characteristics of chalky rice grains generated by ripening under high temperature and compared them with whole grains. We evaluated 14 unpolished Japonica rice grains harvested in Japan in 2021, and these samples (original grains) were divided into two groups (a whole grain group and a chalky grain one). We found that not only activities of endogenous amylase and proteinase, but also cell wall-degrading enzymes, such as xylanase and cellulase, changed markedly between chalky grains and whole grains. Using rice grains blended with 30% of chalky grains as the material, we compared the sugar and mineral contents and textural properties of the rice grains soaked and boiled in either ordinary water or hard water, such as Evian or Contrex. It was shown that xylanase, in addition to amylase and proteinase, may play an important role in changing the texture of the boiled chalky rice grains. For the sake of preventing the above-mentioned deterioration in the texture of boiled grains of chalky rice, we tried to use hard water, such as Evian or Contrex, to soak and cook the chalky rice grains. It was shown that the hard water was useful for the prevention of texture deterioration of the boiled rice grains due to inhibition of the activities of endogenous hydrolytic enzymes, such as α-amylase, β-amylase, proteinase, and xylanase. Furthermore, we found that the hard water was useful in increasing the calcium absorption through the meal by 2.6 to 16.5 times.
Hide AbstractDurum Wheat Bread with a Potentially High Health Value through the Addition of Durum Wheat Thin Bran or Barley Flour.
Ficco, D. B. M., Canale, M., Giannone, V., Strano, M. C., Allegra, M., Zingale, S. & Spina, A. (2023). Plants, 12(2), 397.
The enrichment of semolina bread with prebiotic ingredients such as β-glucans may exert health-promoting effects. This work presents the results of a general recipe development aimed at improving the nutritional value of bakery products. In this study, increasing amounts (0%, 2%, 5%, 7%, and 10%) of thin bran or barley flour were added into re-milled durum wheat semolina to prepare breads. The technological quality of doughs and breads was investigated. In general, the Farinograph water absorption of flour and dough stability increased with increasing inclusion levels of barley flour or thin bran (up to 73.23% and 18.75 min, respectively), contrarily to the increase of dough development time only in barley inclusion (4.55 min). At the same time, the softening index decreased for almost all of these, except for 2% of thin bran or barley flour inclusion. At Mixograph, mixing time increased (up to 5.13 min) whilst the peak height decreased. The specific volume and hardness of loaf differently decreased for almost all thesis (ranges 12.6–24.0% and 39.4–45.5%, respectively). The other quality parameters remained unchanged compared with semolina bread. After baking, β-glucan levels increased differently at all the inclusion levels (2.35-fold, on average). The breadcrumb color was deep brown, while the crust became lighter in color. The breads contain β-glucans even at low percentages of barley/bran inclusions while maintaining their technological performance. In conclusion, the results show an interesting potential of barley flour or thin bran as ingredients in breadmaking to increase the β-glucans daily intake, but further investigations are needed to achieve improved quality features.
Hide AbstractInfluence of sweet potato age and storage on cooking quality parameters.
Somerfield, S. D., Searle, B., Hedderley, D. & O’Donoghue, E. M. (2023). New Zealand Journal of Crop and Horticultural Science, 1-16.
Small-sized sweet potatoes are often sold as a specialty product, graded out from the main harvest, but they can also be intentionally achieved by harvesting much earlier than usual. Since these roots are biologically young, questions arise as to their ability to convert starch to maltose in a similar manner to the older, full-sized roots. Here we show that small sweet potatoes harvested 59 days post-planting had lower dry matter and starch than full-sized sweet potatoes (harvested 120 days post-planting), reflecting the active growth stage of the young root. The concentration of maltose produced during cooking (dry basis) was lower in the young sweet potatoes. However, the proportion of available starch converted to maltose through β-amylase activity was similar (60–65%). An eight-week postharvest storage at 15°C had no effect on β-amylase activity or the concentration of maltose produced on cooking. Total sweetness index (sucrose equivalents) in cooked tissue was lower on a fresh basis for young roots compared to full-sized roots but was not different when expressed on a dry basis, suggesting that the sweetness intensity of young roots could potentially be improved by introducing a water deficit prior to harvest, or cooking practices such as fan baking.
Hide AbstractBiochemical Properties of β-Amylase from Red Algae and Improvement of Its Thermostability through Immobilization.
Murakami, M. & Osanai, T. (2022). ACS omega, 7, 36195-36205.
β-Amylase hydrolyzes polysaccharides, such as starch, into maltose. It is used as an industrial enzyme in the production of food and pharmaceuticals. The eukaryotic red alga Cyanidioschyzon merolae is a unicellular alga that grows at an optimum pH of 2.0-3.0 and an optimum temperature of 40-50°C. By focusing on the thermostability and acid resistance of the proteins of C. merolae, we investigated the properties of β-amylase from C. merolae (hereafter CmBAM) and explored the possibility of using CmBAM as an industrial enzyme. CmBAM showed the highest activity at 47°C and pH 6.0. CmBAM had a relatively higher specificity for amylose as a substrate than for starch. Immobilization of CmBAM on a silica gel carrier improved storage stability and thermostability, allowing the enzyme to be reused. The optimum temperature and pH of CmBAM were comparable to those of existing β-amylases from barley and wheat. C. merolae does not use amylose, but CmBAM has a substrate specificity for both amylose and amylopectin but not for glycogen. Among the several β-amylases reported, CmBAM was unique, with a higher specificity for amylose than for starch. The high specificity of CmBAM for amylose suggests that isoamylase and pullulanase, which cleave the α-1,6 bonds of starch, may act together in vivo. Compared with several reported immobilized plant-derived β-amylases, immobilized CmBAM was comparable to β-amylase, with the highest reusability and the third-highest storage stability at 30 days of storage. In addition, immobilized CmBAM has improved thermostability by 15-20°C, which can lead to wider applications and easier handling.
Hide Abstract