| Content: | 5,000 Units |
| Shipping Temperature: | Ambient |
| Storage Temperature: | 2-8oC |
| Formulation: | In 3.2 M ammonium sulphate |
| Physical Form: | Suspension |
| Stability: | > 1 year under recommended storage conditions |
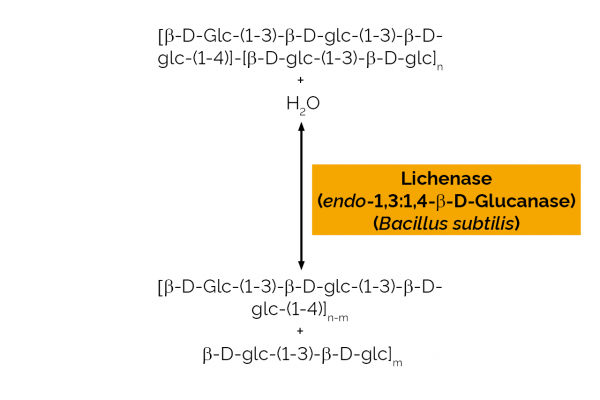
| Enzyme Activity: | β-Glucanase/Lichenase |
| EC Number: | 3.2.1.73 |
| CAZy Family: | GH16 |
| CAS Number: | 37288-51-0 |
| Synonyms: | licheninase; (1→3)-(1→4)-beta-D-glucan 4-glucanohydrolase |
| Source: | Bacillus subtilis |
| Molecular Weight: | 26,750 |
| Concentration: | Supplied at ~ 1,000 U/mL |
| Expression: | Purified from Bacillus subtilis |
| Specificity: | Hydrolysis of (1,4)-β-D-glucosidic linkages in β-D-glucans containing (1,3)- and (1,4)-bonds. |
| Specific Activity: | ~ 250 U/mg (40oC, pH 6.5 on barley β-glucan) |
| Unit Definition: | One Unit of lichenase activity is defined as the amount of enzyme required to release one µmole of glucose reducing-sugar equivalents per minute from barley β-glucan (10 mg/mL) in sodium phosphate buffer (100mM), pH 6.5 at 40oC. |
| Temperature Optima: | 60oC |
| pH Optima: | 6 |
| Application examples: | Applications in carbohydrate research and in the food and feeds, brewing and biofuels industries. |
High purity Lichenase (endo-1,3,1,4-β-Glucanase) (Bacillus subtilis) for use in research, biochemical enzyme assays and in vitro diagnostic analysis.
See all purified Carbohydrate Active enZYmes available.
Mangan, D., Liadova, A., Ivory, R. & McCleary, B. V. (2016). Carbohydrate Research, 435, 162-172.
We report herein the development of a novel assay procedure for the measurement of β-glucanase and lichenase (EC 3.2.1.73) in crude enzyme extracts. Two assay formats based on a) a direct cleavage or b) an enzyme coupled substrate were initially investigated. The ‘direct cleavage’ substrate, namely 4,6-O-benzylidene-2-chloro-4-nitrophenyl-β-31-cellotriosyl-β-glucopyranoside (MBG4), was found to be the more generally applicable reagent. This substrate was fully characterised using a crude malt β-glucanase extract, a bacterial lichenase (Bacillus sp.) and a non-specific endo-1,3(4)-β-glucanase from Clostridium thermocellum (EC 3.2.1.6). Standard curves were derived that allow the assay absorbance response to be directly converted to β-glucanase/lichenase activity on barley β-glucan. The specificity of MBG4 was confirmed by analysing the action of competing glycosyl hydrolases that are typically found in malt on the substrate. Manual and automated assay formats were developed for the analysis of a) β-glucanase in malt flour and b) lichenase enzyme extracts and the repeatability of these assays was fully investigated.
Hide AbstractMcCleary, B. V. & Codd, R. (1991). Journal of the Science of Food and Agriculture, 55(2), 303-312.
A commercially available enzymic method for the quantitative measurement of (1→3),(1→4)-β-glucan has been simplified to allow analysis of up to 10 grain samples in 70 min or of 100–200 samples by a single operator in a day. These improvements have been achieved with no loss in accuracy or precision and with an increase in reliability. The glucose oxidase/peroxidase reagent has been significantly improved to ensure colour stability for periods of up to 1 h after development. Some problems experienced with the original method have been addressed and resolved, and further experiments to demonstrate the quantitative nature of the assay have been designed and performed.
Hide AbstractMcCleary, B. V. & Nurthen, E. (1986). Journal of the Institute of Brewing, 92(2), 168-173.
A method developed for the quantification of (1→3)(1→4)-β-D-glucan in barley flour has been modified to allow its use in the measurement of this component in malt, wort, beer and spent grain. For malt samples, free D-glucose was first removed with aqueous ethanol. Quantification of the polymer in wort and beer samples involved precipitation of the β-glucan with ammonium sulphate followed by washing with aqueous ethanol to remove free D-glucose. Spent grain was lyophilised and milled and then analysed by the method developed for malt. In all cases, the β-glucan was depolymerised with lichenase and the resultant β-gluco-oligosaccharides hydrolysed to D-glucose with β-D-glucosidase. The released D-glucose was then specifically determined using glucose oxidase-peroxidase reagent.
Hide AbstractMcCleary, B. V. & Glennie-Holmes, M. (1985). Journal of the Institute of Brewing, 91(5), 285-295.
A simple and quantitative method for the determination of (1→3) (1→4)-β-D-glucan in barley flour and malt is described. The method allows direct analysis of β-glucan in flour and malt slurries. Mixed-linkage β-glucan is specifically depolymerized with a highly purified (1→3) (1→4)-β-D-glucanase (lichenase), from Bacillus subtilis, to tri-, tetra- and higher degree of polymerization (d.p.) oligosaccharides. These oligosaccharides are then specifically and quantitatively hydrolysed to glucose using purified β-D-glucosidase. The glucose is then specifically determined using glucose oxidase/peroxidase reagent. Since barley flours contain only low levels of glucose, and maltosaccharides do not interfere with the assay, removal of low d.p. sugars is not necessary. Blank values are determined for each sample allowing the direct measurement of β-glucan in values are determined for each sample allowing the direct measurement of β-glucan in malt samples. α-Amylase does not interfere with the assay. The method is suitable for the routine analysis of β-glucan in barley samples derived from breeding programs; 50 samples can be analysed by a single operator in a day. Evaluation of the technique on different days has indicated a mean standard error of 0-1 for barley flour samples containing 3-8 and 4-6% (w/w) β-glucan content.
Hide AbstractCovalent connectivity of glycogen in brewer's spent yeast cell walls revealed by enzymatic approaches and dynamic nuclear polarization NMR.
Bastos, R., Marín-Montesinos, I., Ferreira, S. S., Mentink-Vigier, F., Sardo, M., Mafra, L., Coimbra, M. A. & Coelho, E. (2024). Carbohydrate Polymers, 324, 121475.
Yeast cell walls undergo modifications during the brewing process, leading to a remodelling of their architecture. One significant change is the increased insolubility of the cell wall glycogen pool, likely due to the formation of covalent bonds between glycogen and cell wall polysaccharides. To verify this hypothesis, we extracted the brewer's spent yeast with 4 M KOH, obtaining an insoluble glucan fraction (AE.4 M) primarily composed of (α1 → 4)- and (1 → 3)-linked Glc residues. Dynamic nuclear polarization solid-state NMR of AE.4 M revealed distinct glucan resonances that helped to differentiate between α- and β glucosyl (1 → 4)-linked residues, and confirm covalent linkages between (β1 → 3)-glucans and glycogen through a (β1 → 4)-linkage. The hydrolysis with different endo-glucanases (zymolyase, cellulase, and lichenase) was used to obtain solubilized high molecular weight glycogen fractions. NMR analysis showed that covalent links between glycogen and (β1 → 6)-glucans through (α1 → 6) glycosidic linkage, with branching at the C6 position involving (β1 → 3), and (β1 → 6)-glucans. HPAEC-PAD analysis of the enzymatically released oligosaccharides confirmed covalent linkages of (β1 → 3), (β1 → 6)-, and (β1 → 4)-glucan motifs with (α1 → 4)-glucans. This combination of multiple enzymatic approaches and NMR methods shed light into the role of yeast cell wall glycogen as a structural core covalently linked to other cell wall components during the brewing process.
Hide AbstractThe Gram-positive bacterium Romboutsia ilealis harbors a polysaccharide synthase that can produce (1, 3; 1, 4)-β-D-glucans.
Chang, S. C., Kao, M. R., Saldivar, R. K., Díaz-Moreno, S. M., Xing, X., Furlanetto, V., Yayo, J., Divne, C., Vilaplana, F., Abbott, D. W. & Hsieh, Y. S. (2023). Nature Communications, 14(1), 4526.
(1,3;1,4)-β-D-Glucans are widely distributed in the cell walls of grasses (family Poaceae) and closely related families, as well as some other vascular plants. Additionally, they have been found in other organisms, including fungi, lichens, brown algae, charophycean green algae, and the bacterium Sinorhizobium meliloti. Only three members of the Cellulose Synthase-Like (CSL) genes in the families CSLF, CSLH, and CSLJ are implicated in (1,3;1,4)-β-D-glucan biosynthesis in grasses. Little is known about the enzymes responsible for synthesizing (1,3;1,4)-β-D-glucans outside the grasses. In the present study, we report the presence of (1,3;1,4)-β-D-glucans in the exopolysaccharides of the Gram-positive bacterium Romboutsia ilealis CRIBT. We also report that RiGT2 is the candidate gene of R. ilealis that encodes (1,3;1,4)-β-D-glucan synthase. RiGT2 has conserved glycosyltransferase family 2 (GT2) motifs, including D, D, D, QXXRW, and a C-terminal PilZ domain that resembles the C-terminal domain of bacteria cellulose synthase, BcsA. Using a direct gain-of-function approach, we insert RiGT2 into Saccharomyces cerevisiae, and (1,3;1,4)-β-D-glucans are produced with structures similar to those of the (1,3;1,4)-β-D-glucans of the lichen Cetraria islandica. Phylogenetic analysis reveals that putative (1,3;1,4)-β-D-glucan synthase candidate genes in several other bacterial species support the finding of (1,3;1,4)-β-D-glucans in these species.
Hide AbstractTopography of UV-melanized Thalli of Lobaria pulmonaria (L.) Hoffm.
Daminova, A. G., Rassabina, A. E., Khabibrakhmanova, V. R., Beckett, R. P. & Minibayeva, F. V. (2023). Plants (Basel), 12(14), 2627.
Lichens are unique extremophilic organisms due to their phenomenal resistance to adverse environmental factors, including ultraviolet (UV) irradiation. Melanization plays a special role in the protection of lichens from UV-B stress. In the present study, we analyzed the binding of melanins with the components of cell walls of the mycobiont of the upper cortex in the melanized lichen thalli Lobaria pulmonaria. Using scanning electron and atomic force microscopy, the morphological and nanomechanical characteristics of the melanized layer of mycobiont cells were visualized. Melanization of lichen thalli led to the smoothing of the surface relief and thickening of mycobiont cell walls, as well as the reduction in adhesion properties of the lichen thallus. Treatment of thalli with hydrolytic enzymes, especially chitinase and lichenase, enhanced the yield of melanin from melanized thalli and promoted the release of carbohydrates, while treatment with pectinase increased the release of carbohydrates and phenols. Our results suggest that melanin can firmly bind with hyphal cell wall carbohydrates, particularly chitin and 1,4-β-glucans, strengthening the melanized upper cortex of lichen thalli, and thereby it can contribute to lichen survival under UV stress.
Hide AbstractMixed-linkage (1, 3; 1, 4)-β-D-glucans as rehydration media for improved redispersion of dried cellulose nanofibrils.
Zha, L., Wang, S., Berglund, L. & Zhou, Q. (2022). Carbohydrate Polymers, 120276.
Improving the redispersion and recycling of dried cellulose nanofibrils (CNFs) without compromising their nanoscopic dimensions and inherent mechanical properties are essential for their large-scale applications. Herein, mixed-linkage (1,3;1,4)-β-d-glucan (MLG) was studied as a rehydration medium for the redispersion and recycling of dried CNFs, benefiting from the intrinsic affinity of MLG to both cellulose and water molecules as inspired from plant cell wall. MLG from barley with a lower molar ratio of cellotriosyl to cellotetraosyl units was found homogeneously coated on CNFs, facilitating rehydration of the network of individualized CNFs. The addition of barley MLG did not impair the mechanical properties of the CNF/MLG composites as compared to neat CNFs nanopaper. With the addition of 10 wt% barley MLG, dry CNF/MLG composite film was successfully redispersed in water and recycled with well-maintained mechanical properties, while lichenan from Icelandic moss, cationic starch, and xyloglucan could not help the redispersion of dried CNFs.
Hide AbstractThe Role of Two Linear β-Glucans Activated by c-di-GMP in Rhizobium etli CFN42.
Pérez-Mendoza, D., Romero-Jiménez, L., Rodríguez-Carvajal, M. Á., Lorite, M. J., Muñoz, S., Olmedilla, A. & Sanjuán, J. (2022). Biology, 11(9), 1364.
Bacterial exopolysaccharides (EPS) are secreted biopolymers with often critical roles in bacterial physiology and ecology. In addition to their biological role, there is increasing interest for EPS in various industrial sectors. β-glucans are among the most important ones including cellulose as the most abundant organic polymer on earth, but also newcomers, such as the bacterial Mixed Linkage β-Glucan (MLG), displaying a unique repeating unit suggestive of biotechnological potential. In this work we describe Rhizobium etli as the first bacterium reported to be able to produce these two linear β-glucans cellulose and MLG. Rhizobium etli is an agronomic relevant rhizobacteria able to perform Biological Nitrogen Fixation (BNF) in a symbiotic association with common bean plants. The production and regulation of cellulose and MLG by Rhizobium etli CFN42 is discussed and their impact on its free-living and symbiotic lifestyles evaluated.
Hide AbstractMicrobial liberation of N-methylserotonin from orange fiber in gnotobiotic mice and humans.
Han, N. D., Cheng, J., Delannoy-Bruno, O., Webber, D., Terrapon, N., Henrissat, B., et al. (2022). Cell, 185(14), 2495-2509.
Plant fibers in byproduct streams produced by non-harsh food processing methods represent biorepositories of diverse, naturally occurring, and physiologically active biomolecules. To demonstrate one approach for their characterization, mass spectrometry of intestinal contents from gnotobiotic mice, plus in vitro studies, revealed liberation of N-methylserotonin from orange fibers by human gut microbiota members including Bacteroides ovatus. Functional genomic analyses of B. ovatus strains grown under permissive and non-permissive N-methylserotonin “mining” conditions revealed polysaccharide utilization loci that target pectins whose expression correlate with strain-specific liberation of this compound. N-methylserotonin, orally administered to germ-free mice, reduced adiposity, altered liver glycogenesis, shortened gut transit time, and changed expression of genes that regulate circadian rhythm in the liver and colon. In human studies, dose-dependent, orange-fiber-specific fecal accumulation of N-methylserotonin positively correlated with levels of microbiome genes encoding enzymes that digest pectic glycans. Identifying this type of microbial mining activity has potential therapeutic implications.
Hide Abstract