| Content: | 55 assays per kit |
| Shipping Temperature: | Ambient |
| Storage Temperature: |
Short term stability: 2-8oC, Long term stability: See individual component labels |
| Stability: | > 2 years under recommended storage conditions |
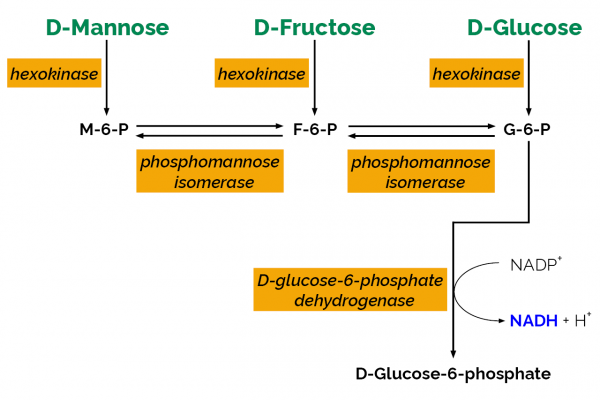
| Analyte: | D-Fructose, D-Glucose, D-Mannose |
| Assay Format: | Spectrophotometer |
| Detection Method: | Absorbance |
| Wavelength (nm): | 340 |
| Signal Response: | Increase |
| Linear Range: | 4 to 80 µg of D-glucose, D-fructose or D-mannose per assay |
| Limit of Detection: | ~ 0.7 mg/L |
| Reaction Time (min): | ~ 30 min |
| Application examples: | Foodstuffs, yeast cell preparations, enzymatic hydrolysates and other materials (e.g. biological cultures, samples, etc.). |
| Method recognition: | Novel method |
The D-Mannose/D-Fructose/D-Glucose test kit is suitable for the specific measurement and analysis of D-mannose, D-fructose and D-glucose in plant products and in acid hydrolysates of polysaccharides.
Note for Content: The number of manual tests per kit can be doubled if all volumes are halved. This can be readily accommodated using the MegaQuantTM Wave Spectrophotometer (D-MQWAVE).
See our full range of monosaccharide and disaccharide assay kits.

- Very competitive price (cost per test)
- All reagents stable for > 2 years after preparation
- Only enzymatic kit available
- Simple format
- Rapid reaction
- Mega-Calc™ software tool is available from our website for hassle-free raw data processing
- Standard included
Influence of grape quality tier, harvest timing, and yeast strain on mannoprotein content, phenolic composition, and color modulation in young red wines.
Galaz-Torres, C., Vidal, J., Vargas, S., Zincker, J., Brossard, N., Bordeu, E., Ricci, A., Parpinello, G. P. & Versari, A. (2025). Food Research International, 205, 115956.
Besides the impact of harvest timing and grape quality, this study investigated the performance of selected yeast strains overproducing mannoproteins versus the conventional strain AGL 804 in terms of mannoprotein release and its consequences on the composition and color modulation of short-aged commercial red wines. The study fills a gap of volume and actual red wine production by comparing 36 winemaking conditions, each of 850 kg of grapes. The results showed that AGL 804 produced the same or more concentration of mannoproteins than the alternatives. Then, an apparent threshold was found for all yeasts when the dissolved solids content of grape musts at harvest exceeded 22 °Bx, beyond which no increase in mannoprotein production was observed. Only below this limit, an independent effect of yeast strains on tannin concentration and tannin-to-anthocyanin ratio (T/A) in wines was observed. These two parameters exhibited a moderate correlation with mannoprotein concentration (R2 = 0.534 and R2 = 0.696, respectively), and a low-moderate correlation for mannoprotein concentration with CIELAB color parameters. Wines produced from grapes > 22 °Bx showed only harvest-related variations in tannin concentration and T/A and no correlation between parameters analyzed and mannoprotein production. The study revealed that, although yeast strains influence the color of red wine after six months of bottling, their effect is secondary to the harvest timing. Moreover, the influence of the yeast strain itself was not consistent across the different harvest dates. Additionally, the study provides winemakers with an improved and practical assay for measuring mannoprotein levels in red wines, especially for small winery laboratories. It also introduces a novel 3D graphical representation of the CIELAB color parameters, which simultaneously integrates the real visible color of the wines and its visual discriminability to the human eye.
Hide AbstractSequential extraction optimization of compounds of interest from spent brewer's yeast biomass treated by Pulsed Electric Fields.
Berzosa, A., Marín-Sánchez, J., Delso, C., Sanz, J., Álvarez, I., Sánchez-Gimeno, C. & Raso, J. (2024). Innovative Food Science & Emerging Technologies, 94, 103705.
The brewing industry produces significant volumes of spent brewer's yeast (SBY), which presents an intriguing opportunity for valorization. This study aims to optimize the extraction of various compounds of interest from electroporated SBY located both in the cytoplasm (amino acids, glutathione and proteins) and in the cell walls (mannoproteins). The optimization of the extraction time, temperature and pH, allowed obtaining an extract rich in glutathione of 2.31 ± 0.15 mg/g dw after 1 h of incubation (pH 8; 30°C) and, a second extract rich in amino acids (155.74 ± 7.83 mg/g dw) and proteins (331.70 ± 15.64 mg/g dw) after a second incubation (37°C, 47 h) of the biomass. To achieve comprehensive valorization of SBY, the exhausted yeast biomass was incubated with lyticase to extract mannoproteins from the cell wall. This study showcases the efficacy of a multiple response function in optimizing valuable compound extraction from electroporated SBY, aligning with circularity principles.
Hide AbstractEffect of a Bacillus subtilis plus Yeast Cell Wall Synbiotic on Salmonella Enteritidis Colonization in Ceca of Layer Pullets.
Araba, M., Girgis, G., McBride, H. & Lohrmann, T. (2024). Poultry, 3(1), 26-35.
Salmonella Enteritidis (SE) is a major contamination concern in eggs and risk for Salmonellosis in humans. Strains of Bacillus subtilis and yeast cell wall can be used as substitutes for antibiotic substances in feed against Salmonella in poultry. The objective of this study was to assess the effect of BacPack® (Quality Technology International, Inc., Elgin, IL, USA) Q1+1 (BPQ11), a feed additive combination of a Bacillus subtilis strain and Saccharomyces cerevisiae cell wall, on SE cecal colonization in Lohmann LSL pullets. A control group (CON) and a test group (BPQ) were each randomly assigned 100-day-old chicks. CON was fed a corn–soybean meal-based vegetarian mash diet, and BPQ was fed the control diet supplemented with BPQ11 for the duration of the study. At 8 days of age, chicks were orally challenged with a nalidixic acid-resistant SE strain at a dose of 6.3 × 107 colony forming units (CFUs) per bird. At 7, 11, 15, and 19 days post-challenge (DPC), 25 birds per group were euthanized, and their cecal contents were collected and analyzed for SE. SE counts were 6.88, 7.98, 7.79, and 7.50 in CON and 7.18, 7.31, 6.35, and 6.30 log10 CFU/g in BPQ at 7, 11, 15, and 19 DPC, respectively. SE did not differ between CON and BPQ at 7 DPC; however, BPQ had lower (p < 0.0001) SE at 11 (−0.67), 15 (−1.45), and 19 (−1.20 log10 CFU/g) DPC. Results indicate that synbiotic BPQ11 may be a useful dietary pre-harvest tool for SE management in layer birds.
Hide AbstractStructural and functional characterization of a multi-domain GH92 α-1, 2-mannosidase from Neobacillus novalis.
Kołaczkowski, B. M., Moroz, O. V., Blagova, E., Davies, G. J., Møller, M. S., Meyer, A. S., Westh, K., Jensen, K., Wilson, K. S. & Krogh, K. B. (2023). Acta Crystallographica Section D: Structural Biology, 79(5), 387-400.
Many secreted eukaryotic proteins are N-glycosylated with oligosaccharides composed of a high-mannose N-glycan core and, in the specific case of yeast cell-wall proteins, an extended α-1,6-mannan backbone carrying a number of α-1,2- and α-1,3-mannose substituents of varying lengths. α-Mannosidases from CAZy family GH92 release terminal mannose residues from these N-glycans, providing access for the α-endomannanases, which then degrade the α-mannan backbone. Most characterized GH92 α-mannosidases consist of a single catalytic domain, while a few have extra domains including putative carbohydrate-binding modules (CBMs). To date, neither the function nor the structure of a multi-domain GH92 α-mannosidase CBM has been characterized. Here, the biochemical investigation and crystal structure of the full-length five-domain GH92 α-1,2-mannosidase from Neobacillus novalis (NnGH92) with mannoimidazole bound in the active site and an additional mannoimidazole bound to the N-terminal CBM32 are reported. The structure of the catalytic domain is very similar to that reported for the GH92 α-mannosidase Bt3990 from Bacteroides thetaiotaomicron, with the substrate-binding site being highly conserved. The function of the CBM32s and other NnGH92 domains was investigated by their sequential deletion and suggested that whilst their binding to the catalytic domain was crucial for the overall structural integrity of the enzyme, they appear to have little impact on the binding affinity to the yeast α-mannan substrate. These new findings provide a better understanding of how to select and optimize other multi-domain bacterial GH92 α-mannosidases for the degradation of yeast α-mannan or mannose-rich glycans.
Hide AbstractIdentification and characterization of an α-1, 3 mannosidase from Elizabethkingia meningoseptica and its potential attenuation impact on allergy associated with cross-reactive carbohydrate determinant.
Shen, D., Lu, X., Li, W., Zou, L., Tong, Y., Wang, L., Rao, L., Zhang, Y, Hou, L., Sun, G, & Chen, L. (2023). Biochemical and Biophysical Research Communications. (2023). Journal of Biochemical and Biophysical Research communications, 672, 17-26.
Core α-1,3 mannose is structurally near the core xylose and core fucose on core pentasaccharide from plant and insect glycoproteins. Mannosidase is a useful tool for characterization the role of core α-1,3 mannose in the composition of glycan related epitope, especially for those epitopes in which core xylose and core fucose are involved. Through functional genomic analysis, we identified a glycoprotein α-1,3 mannosidase and named it MA3. We used MA3 to treat allergen horseradish peroxidase (HRP) and phospholipase A2 (PLA2) separately. The results showed that after MA3 removed α-1,3 mannose on HRP, the reactivity of HRP with anti-core xylose polyclonal antibody almost disappeared. And the reactivity of MA3-treated PLA2 with anti-core fucose polyclonal antibody decreased partially. In addition, when PLA2 was conducted enzyme digestion by MA3, the reactivity between PLA2 and allergic patients’ sera diminished. These results demonstrated that α-1,3 mannose was an critical component of glycan related epitope.
Hide AbstractAnalysis of the galactomannan binding ability of β-mannosidases, BtMan2A and CmMan5A, regarding their activity and synergism with a β-mannanase.
Malgas, S., Thoresen, M., Moses, V., Prinsloo, E., van Dyk, J. S. & Pletschke, B. I. (2022). Computational and Structural Biotechnology Journal, 20, 3140-3150.
Both β-mannanases and β-mannosidases are required for mannan-backbone degradation into mannose. In this study, two β-mannosidases of glycoside hydrolase (GH) families 2 (BtMan2A) and 5 (CmMan5A) were evaluated for their substrate specificities and galactomannan binding ability. BtMan2A preferred short manno-oligomers, while CmMan5A preferred longer ones; DP >2, and galactomannans. BtMan2A displayed irreversible galactomannan binding, which was pH-dependent, with higher binding observed at low pH, while CmMan5A had limited binding. Docking and molecular dynamics (MD) simulations showed that BtMan2A galactomannan binding was stronger under acidic conditions (-8.4 kcal/mol) than in a neutral environment (-7.6 kcal/mol), and the galactomannan ligand was more unstable under neutral conditions than acidic conditions. Qualitative surface plasmon resonance (SPR) experimentally confirmed the reduced binding capacity of BtMan2A at pH 7. Finally, synergistic β-mannanase to β-mannosidase (BtMan2A or CmMan5A) ratios required for maximal galactomannan hydrolysis were determined. All CcManA to CmMan5A combinations were synergistic (≈1.2-fold), while combinations of CcManA with BtMan2A (≈1.0-fold) yielded no hydrolysis improvement. In conclusion, the low specific activity of BtMan2A towards long and galactose-containing oligomers and its non-catalytic galactomannan binding ability led to no synergy with the mannanase, making GH2 mannosidases ineffective for use in cocktails for mannan degradation.
Hide AbstractThe protective effect of Scenedesmus dimorphus polysaccharide as an antioxidant and antiaging agent on aging rat model induced by D-galactose.
Armaini, A. & Imelda, I. (2021). Journal of Applied Pharmaceutical Science, 11(05), 054-063.
Premature skin aging occurs due to the increased formation of reactive oxygen species (ROS), which causes oxidative stress, DNA damage, and collagen degradation. This study investigates the protective effect of Scenedesmus dimorphus polysaccharides (SDP) as an antioxidant and anti-aging agent on an aging rat model induced by D-galactose (D-gal). This study used 48 male Wistar rats divided into six groups: (1) normal control, (2) polysaccharide control, (3) aging control induced by D-gal 0.25 mg/g bw/days, (4) drug control (vitamin E) treated twice with SDP in an aging rat model, (5) D-gal + SDP (0.2 mg/g bw), and (6) D-gal + SDP (0.8 mg/g bw) treated with oral treatment and observed for periods of 2, 4, and 8 weeks. Giving SDP at a dose of 0.8 mg/g bw can increase superoxide dismutases and catalase activity and reduce malondialdehyde after 8 weeks of observation, which is better than giving vitamin E. The treatment of SDP can stimulate collagen synthesis and reduce advanced glycation end products. Histopathology shows an increase in the area of fibrocollagen proportions and deposition from the matrix on giving SDP, which is better than vitamin E, since SDP can repair skin tissue. Thus, SDP can be useful as an antioxidant and anti-aging agent in an aging Wistar rats model.
Hide AbstractEvolution of mutualistic behaviour between Chlorella sorokiniana and Saccharomyces cerevisiae within a synthetic environment.
Oosthuizen, J. R., Naidoo, R. K., Rossouw, D. & Bauer, F. F. (2020). Journal of Industrial Microbiology & Biotechnology, 47(4-5), 357-372.
Yeast and microalgae are microorganisms with widely diverging physiological and biotechnological properties. Accordingly, their fields of applications diverge: yeasts are primarily applied in processes related to fermentation, while microalgae are used for the production of high-value metabolites and green technologies such as carbon capture. Heterotrophic–autotrophic systems and synthetic ecology approaches have been proposed as tools to achieve stable combinations of such evolutionarily unrelated species. We describe an entirely novel synthetic ecology-based approach to evolve co-operative behaviour between winery wastewater isolates of the yeast Saccharomyces cerevisiae and microalga Chlorella sorokiniana. The data show that biomass production and mutualistic growth improved when co-evolved yeast and microalgae strains were paired together. Combinations of co-evolved strains displayed a range of phenotypes, including differences in amino acid profiles. Taken together, the results demonstrate that biotic selection pressures can lead to improved mutualistic growth phenotypes over relatively short time periods.
Hide AbstractImmunotherapy based on Pythium insidiosum mycelia drives a Th1/Th17 response in mice.
Tondolo, J. S., Loreto, E. S., de Jesus, F. P., Ledur, P. C., Verdi, C. M., & Santurio, J. M. (2020). Medical Mycology, 58(8), 1120-1125.
Pythium insidiosum is an oomycete that affects mammals, especially humans and horses, causing a difficult-to-treat disease. Typically, surgical interventions associated with antimicrobial therapy, immunotherapy, or both are the preferred treatment choices. PitiumVac® is a therapeutic vaccine prepared from the mycelial mass of P. insidiosum and is used to treat Brazilian equine pythiosis. To better understand how PitiumVac® works, we analyzed the composition of PitiumVac® and the immune response triggered by this immunotherapy in mice. We performed an enzymatic quantification that showed a total glucan content of 21.05% ± 0.94 (α-glucan, 6.37% ± 0.77 and (1,3)(1,6)-β-glucan, 14.68% ± 0.60) and mannose content of 1.39% ± 0.26; the protein content was 0.52 mg ml−1 ± 0.07 mg ml−1. Healthy Swiss mice (n = 3) were subcutaneously preimmunized with one, two, or three shots of PitiumVac®, and immunization promoted a relevant Th1 and Th17 responses compared to nonimmunization of mice. The highest cytokine levels were observed after the third immunization, principally for IFN-γ, IL-17A, IL-6, and IL-10 levels. Results of infected untreated (Pythiosis) and infected treated (Pythiosis + PVAC) mice (n = 3) showed that PitiumVac® reinforces the Th1/Th17 response displayed by untreated mice. The (1,3)(1,6)-β-glucan content can be, at least in part, related to this Th1/Th17 response.
Hide AbstractCombined Yeast Cultivation and Pectin Hydrolysis as an Effective Method of Producing Prebiotic Animal Feed from Sugar Beet Pulp.
Wilkowska, A., Berlowska, J., Nowak, A., Motyl, I., Antczak-Chrobot, A., Wojtczak, M., Kunicka-Styczyńska, A., Binczarski, M. & Dziugan, P. (2020). Biomolecules, 10(5), 724.
An effective and ecological method for liberation of pectin-derived oligosaccharides (POS) from sugar beet pulp (SBP) was developed using enzymatic and microorganism-mediated biomass conversion. The POS may be applied in the production of prebiotic feed additives. Various yeast strains were screened for their capacity for protein synthesis and monosaccharide assimilation. Combined yeast cultivation and pectin hydrolysis were found to be an effective method of producing prebiotics. Separate enzymatic hydrolysis and fermentation of SBP resulted in the release of 3.6 g of POS per 100 g d.w., whereas the yield of POS acquired after the combined process was 17.9% higher, giving 4.2 g of POS per 100 g d.w. Introducing the yeast into the process improved hydrolysis performance due to lower enzyme inhibition by mono- and disaccharides. The prebiotic effect of the POS was assessed by in vitro fermentation using individual cultures of gastrointestinal bacteria. The POS in the SBP hydrolysate effectively promoted the growth of lactobacilli and bifidobacteria. A large increase in adherence to Caco-2 cells in the presence of POS was noted for beneficial Lactobacillus brevis strains, whereas pathogenic bacteria and yeast (C. albicans, C. lusitanie, C. pelliculosa), responsible for infections in breeding animals, showed much weaker adhesion.
Hide AbstractLysates of Metschnikowia Yeast with Higher Content of Hydroxyproline.
Pawlikowska, E., Szymanska, M., Berlowska, J. & Kregiel, D. (2020). BioResources, 15(2), 3228-3236.
The chemical characteristics of lysates obtained from yeasts belonging to Metschnikowia spp. were determined. Cell lysis was induced using saponin from Q. saponaria or 5% NaCl. The process was conducted at 50°C for 24 for 48 h. The enzymatic profiles of the resulting lysates were analyzed. The mannose and glucose contents were also investigated, as well as the concentrations of proteins, free amino nitrogen (FAN), and free amino acids. The results were compared to the characteristics of lysates from conventional industrial strains of Saccharomyces spp. obtained under analogous conditions. The Metschnikowia lysates showed different chemical profiles and the pool of individual amino acids was generally smaller. However, the content of hydroxyproline HPro was 4 to 5 times higher. The results of this study show that yeast lysates are an attractive supplement for numerous applications.
Hide AbstractEffect of pulsed electric fields on mannoproteins release from Saccharomyces cerevisiae during the aging on lees of Caladoc red wine.
Maza, M. A., Delso, C., Álvarez, I., Raso, J. & Martínez, J. M. (2020). LWT, 118, 108788.
The potential of PEF for accelerating the release of mannoproteins from Saccharomyces cerevisiae yeast during aging on lees of Caladoc red wine was evaluated. The release of mannoproteins increased rapidly in red wine samples containing PEF-treated yeasts in comparison to samples containing untreated yeast. While one month of aging on lees was required to obtain the highest mannoprotein concentration in wines containing PEF-treated yeast, the same amount of mannoproteins was obtained after three months in the wine-containing untreated yeast. It was demonstrated that PEF treatment promoted the liberation of enzymes involved in the release of mannoproteins from the cell wall of yeast during autolysis such as β-glucanase and protease. The functional properties of mannoproteins released from PEF-treated yeast were similar to that of those released from untreated yeast. Mannoproteins reduced wine astringency. Sensorial analysis confirmed that panelists did not find differences in astringency when wine aged on lees for 1 month with PEF-treated yeast was compared with wine aged on lees for 3 months with untreated yeast. Therefore, PEF represents a potential technique for reducing the duration of aging on lees in red winemaking.
Hide AbstractVegetable wastes derived polysaccharides as natural eco-friendly plasticizers of sodium alginate.
Di Donato, P., Taurisano, V., Poli, A., d’Ayala, G. G., Nicolaus, B., Malinconinco, M. & Santagata, G. (2020). Carbohydrate polymers, 229, 115427.
In this paper, lemon and fennel wastes were recovered and used as secondary-raw polysaccharide sources. These polysaccharides were exploited as natural plasticizers of sodium alginate (A) based films, in order to improve sodium alginate performances, limited by its fragility, extending its potential application in a cost effective and eco-friendly way. Different green processes, such as maceration (MAC), ultrasound assisted extraction (UAE) and microwave assisted extraction (MAE), were carried out for obtaining high yield of lemon and fennel polysaccharides (LP and FP). Actually, HPAE-PAD and TLC analyses evidenced the presence of xyslose, galactose, glucose and rhamnose monomers and galacturonic acid, typical of polysaccharides like pectin and xyloglucan chains. These findings were confirmed by NMR and FTIR spectroscopic analyses. Moreovers, gel filtration chromatography assessed the high molecular weight of recovered polysaccharides, particularly of FP waste fraction. The extracted polysaccharides were used as eco-friendly and cost-effective plasticizers of sodium alginate films (AFP and ALP). DSC analysis evidenced a significant decreasing of glass transition temperature of the polymer, tensile tests showed an enlightened rising of elongation at break and TGA analysis showed a faster degradation kinetics of AFP and ALP films, as expected in a plasticized system.
Hide AbstractCross-linking of diluted alkali-soluble pectin from apple (Malus domestica fruit) in different acid-base conditions.
Gawkowska, D., Cieśla, J., Zdunek, A. & Cybulska, J. (2019). Food Hydrocolloids, 92, 285-292.
A diluted alkali-soluble pectin (DASP) fraction, extracted using sodium carbonate, is characterized by a low degree of methylesterification and has the ability to self-organize on mica. The aim of this study was to characterize the cross-linking process of this fraction, extracted from apples, over a wide pH range (3-11) and without the addition of salt. An FT-IR study showed an increase in the intensity of bands connected with νas and νs (COO−) and a decrease in the intensity of the band associated with ν (C=O) in the carboxyl group with increasing pH, which indicated the dissociation of the carboxyl groups of galacturonic acid units. An increase in the surface electrical charge of particles in the pH range of 3-7 confirmed this. The value of the average apparent dissociation constant (∼4.60) indicated the acidic character of the DASP fraction. An AFM study showed the morphological changes of the DASP fraction with increasing pH, which allowed for the evaluation of the cross-linking process. This fraction formed a network on mica at pH 4 and 9, while the aggregates were noted mainly at pH 11. For totally ionized carboxyl groups (pH 7), the pectin chains were separated from each other due to the electrostatic repulsion between them.
Hide AbstractA Bacteroidetes locus dedicated to fungal 1,6-β-glucan degradation: unique substrate conformation drives specificity of the key endo-1,6-β-glucanase.
Temple, M. J., Cuskin, F., Baslé, A., Hickey, N., Speciale, G., Williams, S. J., Gilbert, H. J. & Lowe, E. C. (2017). Journal of Biological Chemistry, jbc-M117.
Glycans are major nutrients available to the human gut microbiota (HGM). The Bacteroides are generalist glycan degraders and this function is mediated largely by polysaccharide utilization loci (PULs). The genomes of several Bacteroides species contain a PUL, PUL1,6-beta;-glucan, that was predicted to target mixed linked plant 1,3;1,4-beta-glucans. To test this hypothesis we characterized the proteins encoded by this locus in Bacteroides thetaiotaomicron, a member of the HGM. We show here that PUL1,6-β-glucan does not orchestrate the degradation of a plant polysaccharide but targets a fungal cell wall glycan, 1,6-beta-glucan, which is a growth substrate for the bacterium. The locus is upregulated by 1,6-beta-glucan, and encodes two enzymes, a surface endo-1,6-beta-glucanase, BT3312, and a periplasmic beta-glucosidase that targets primarily 1,6-beta-glucans. The non-catalytic proteins encoded by PUL1,6-beta-glucan target 1,6-beta-glucans and comprise a surface glycan binding protein and a SusD homologue that delivers glycans to the outer membrane transporter. We identified the central role of the endo-1,6-beta-glucanase in 1,6-beta-glucan depolymerization by deleting bt3312, which prevented the growth of B. thetaiotaomicron on 1,6-beta-glucan. The crystal structure of BT3312 in complex with β-glucosyl-1,6-deoxynojirimycin, revealed a TIM barrel catalytic domain that contains a deep substrate binding cleft tailored to accommodate the hook-like structure adopted by 1,6-beta-glucan. Specificity is driven by the complementarity of the enzyme active site cleft and the conformation of the substrate. We also noted that PUL1,6-beta-glucan is syntenic to many PULs from other Bacteroidetes suggesting that utilization of yeast and fungal cell wall 1,6-beta-glucans is a widespread adaptation within the human microbiota.
Hide Abstract