180 / 360 assays per kit
This product has been discontinued
| Content: | 180 / 360 assays per kit |
| Shipping Temperature: | Ambient |
| Storage Temperature: |
Short term stability: 2-8oC, Long term stability: See individual component labels |
| Stability: | > 2 years under recommended storage conditions |
| Analyte: | endo-Cellulase |
| Assay Format: | Spectrophotometer, Auto-analyser |
| Detection Method: | Absorbance |
| Wavelength (nm): | 400 |
| Signal Response: | Increase |
| Limit of Detection: | 0.05 U/mL |
| Reproducibility (%): | ~ 3% |
| Total Assay Time: | ~ 20 min |
| Application examples: | Fermentation broths, industrial enzyme preparations and biofuels research. |
| Method recognition: | Novel method |
This product has been discontinued (read more).
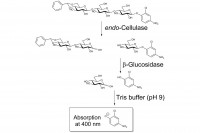
The CellG3 assay reagent for the measurement of endo-cellulase (endo-1,4-β-glucanase) contains two components;
1) 4,6-O-benzylidene-2-chloro-4-nitrophenyl-β-D-cellotrioside (BCNPG3) and 2) thermostable β-glucosidase. The benzylidene blocking group prevents any hydrolytic action by the β-glucosidase on BCNPG3. Incubation with an endo-cellulase generates a non-blocked colourimetric oligosaccharide that is rapidly hydrolysed by the ancillary β-glucosidase. The rate of formation of 2-chloro-4-nitrophenol is therefore directly related to the hydrolysis of BCNPG3 by the endo-cellulase. The reaction is terminated and the phenolate colour is developed on addition of Tris buffer solution (pH 9.0).
Please note that a new assay kit (K-CellG5) is now available for the measurement of endo-cellulase. The CellG5 reagent contains a cellopentaose core and exhibits vastly improved sensitivity for some cellulases. In addition, the exchange of the benzylidene blocking group in CellG3 for 3-keto-butylidene in CellG5 improves the substrate’s water solubility significantly, allowing for a reduction in the concentration of DMSO required in the assay. As DMSO is known to inhibit certain cellulases, this is another benefit in using CellG5. Megazyme now recommends the use of K-CellG5 for all assays for the measurement of endo-cellulase.
Browse more cellulase and other enzyme activity assay kits.

- Very cost effective
- All reagents stable for > 2 years after preparation
- Completely specific for cellulase (endo-1,4-glucanase). The substrate is not hydrolysed by β-glucosidase, cellbiohyrolase or any other enzymes tested
- Kinetic assays possible due to significant phenolate ion presence (and UV absorbance) at pH 5-6
- Simple format. Well suited to automation
- Standard included
A novel automatable enzyme-coupled colorimetric assay for endo-1,4-β-glucanase (cellulase).
Mangan, D., Cornaggia, C., McKie, V., Kargelis. T. & V. McCleary, B. V. (2016). Analytical and Bioanalytical Chemistry, 408(15), 4159-4168.
endo-1,4-β-Glucanase (endo-cellulase, EC 3.2.1.4) is one of the most widely used enzymes in industry. Despite its importance, improved methods for the rapid, selective, quantitative assay of this enzyme have been slow to emerge. In 2014, a novel enzyme-coupled assay that addressed many of the limitations of the existing assay methodology was reported. This involved the use of a bifunctional substrate chemically derived from cellotriose. Reported herein is a much improved version of this assay employing a novel substrate, namely 4,6-O-(3-ketobutylidene)-4-nitrophenyl-β-D-cellopentaoside.
Hide AbstractMcCleary, B. V., Mangan, D., Daly, R., Fort, S., Ivory, R. & McCormack, N. (2014). Carbohydrate Research, 385, 9-17.
A specific and sensitive substrate for the assay of endo-1,4-β-glucanase (cellulase) has been prepared. The substrate mixture comprises benzylidene end-blocked 2-chloro-4-nitrophenyl-β-cellotrioside (BzCNPG3) in the presence of thermostable β-glucosidase. Hydrolysis by exo-acting enzymes such as β-glucosidase and exo-β-glucanase is prevented by the presence of the benzylidene group on the non-reducing end D-glucosyl residue. On hydrolysis by cellulase, the 2-chloro-4-nitrophenyl-β-glycoside is immediately hydrolysed to 2-chloro-4-nitrophenol and free D-glucose by the β-glucosidase in the substrate mixture. The reaction is terminated and colour developed by the addition of a weak alkaline solution. The assay procedure is simple to use, specific, accurate, robust and readily adapted to automation. This procedure should find widespread applications in biomass enzymology and in the specific assay of endo-1,4-β-glucanase in general.
Hide AbstractMangan, D., McCleary, B. V., Liadova, A., Ivory, R. & McCormack, N. (2014). Carbohydrate Research, 395, 47-51.
There is a growing demand for research tools to aid the scientific community in the search for improved cellulase enzymes for the biofuel industry. In this work, we describe a novel fluorometric assay for cellulase (endo-1,4-β-glucanase) which is based on the use of 4,6-O-benzylidene-4-methylumbelliferyl-β-cellotrioside (BzMUG3) in the presence of an ancillary β-glucosidase. This assay can be used quantitatively over a reasonable linear range, or qualitatively as a solution screening tool which may find extensive use in the area of metagenomics.
Hide AbstractEthylene treatment of “Maekawa‐Jiro” persimmon affects peel characteristics and consequently, enables boil‐peeling.
Murakami, S., Yamaguchi, K. & Hashimoto, N. (2021). Food Science & Nutrition, 9(5), 2597-2604.
In a previous study, we reported that ethylene treatment facilitated boil-peeling in persimmons and in several other fruits; however, the mechanism underlying the facilitating effect of ethylene was not examined in detail. Thus, in this study, we investigated the effect of ethylene treatment on the peel characteristics of persimmons, that facilitated boil-peeling, using chemical, genomic, and histochemistry analyses. The results of the study showed that the ethylene-related genes, DK-ACS1 and DK-ACO2, and the pectinase-active gene DKPG were not expressed, even though a minor increase in ethylene generation was observed after ethylene treatment. Conversely, significant accumulation of toluidine blue O and ruthenium red dyes were observed in the sarcocarp and exocarp of the fruits, indicating an increase in the quantity of polysaccharides, including pectic substances, at the site. The results also indicate that the increased cellulase activity observed in the pericarp of the fruits may be due to the aging of the fruits, and not necessarily as a result of ethylene treatment. Furthermore, ethylene treatment increased the quantity of polysaccharides, including pectic substances, directly below the pericarp, which caused the dissolution of the site, resulting in peeling. This study provides new insights on the effect of ethylene on boil-peeling in persimmons and provides a foundation for future research studying the effect of heat treatment in the peeling of fruits or tomato.
Hide AbstractIde, M., Okumura, M., Koizumi, K., Kumagai, M., Yoshida, I., Yoshida, M., Mishima, T. & Nakamura, M. (2018). Journal of Agricultural and Food Chemistry, In Press.
Some β-glucans have attracted attention due to their functionality as an immunostimulant and have been used in processed foods. However, accurately measuring the β-glucan content of processed foods using existing methods is difficult. We demonstrate a new method, the Sodium hypochlorite Extracting and Enzymatic Digesting (SEED) assay, in which β-glucan is extracted using sodium hypochlorite, dimethyl sulfoxide, and 5 mol/L sodium hydroxide and then digested into β-glucan fragments using Westase which is an enzyme having β-1,6- and β-1,3 glucanase activity. The β-glucan fragments are further digested into glucose using exo-1,3-β-D-glucanase and β-glucosidase. We measured β-glucan comprising β-1,3-, -1,6-, and -1,(3),4- bonds in various polysaccharide reagents and processed foods using our novel method. The SEED assay was able to quantify β-glucan with good reproducibility, and the recovery rate was >90% for food containing β-glucan. Therefore, the SEED assay is capable of accurately measuring the β-glucan content of processed foods.
Hide Abstract