170 Units Sucrase plus 3,000 Units β-Galactosidase
This product has been discontinued
| Content: | 170 Units Sucrase plus 3,000 Units β-Galactosidase |
| Shipping Temperature: | Ambient |
| Storage Temperature: | Below -10oC |
| Formulation: | Freeze-dried powder |
| Physical Form: | Powder |
| Stability: | > 1 year under recommended storage conditions |
| Enzyme Activity: | β-Galactosidase, Sucrase/Invertase |
| EC Number: |
Sucrase: 3.2.1.20, β-Galactosidase: 3.2.1.23 |
| CAZy Family: | GH13, GH35 |
| CAS Number: |
Sucrase: 9001-42-7, β-Galactosidase: 9031-11-2 |
| Synonyms: |
Sucrase: alpha-glucosidase; alpha-D-glucoside glucohydrolase, β-Galactosidase: beta-galactosidase; beta-D-galactoside galactohydrolase |
| Source: |
Sucrase: Yeast, β-Galactosidase: Aspergillus niger |
| Molecular Weight: | 57,750 |
| Concentration: | Sucrase (170 U) / β-Galactosidase (3,000 U) |
| Expression: |
Sucrase from Yeast. β-Galactosidase from Aspergillus niger. |
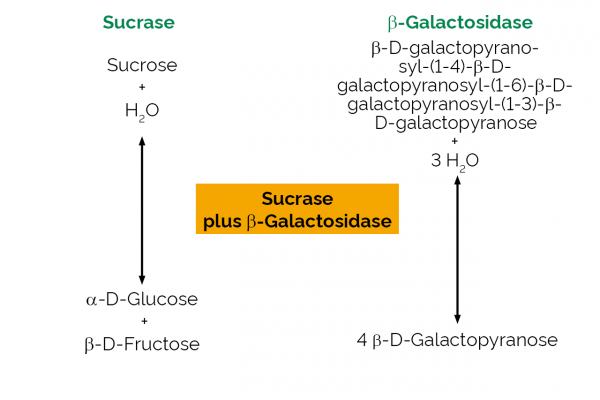
| Specificity: | This enzyme mixture gives complete hydrolysis of sucrose, maltose and lactose under the defined assay conditions, with no hydrolysis of the trisaccharide, β-D-Fruf-(2,1)-β-D-Fruf-(2,1)-β-D-Fruf. |
| Specific Activity: | Sucrase (170 U) / β-Galactosidase (3,000 U) |
| Unit Definition: |
Sucrase: One Unit of sucrase activity is defined as the amount of enzyme required to release one µmole of glucose per minute from sucrose (10 mM) in sodium phosphate buffer (100 mM), pH 6.8 at 40oC. β-Galactosidase: One Unit of β-galactosidase activity is defined as the amount of enzyme required to release one µmole of p-nitrophenol (pNP) per minute from p-nitrophenyl-β-D-galactoside (10 mM) in sodium acetate buffer (100 mM), pH 4.5 at 40oC. |
| Temperature Optima: | 40oC |
| pH Optima: | 5 |
| Application examples: | Used for removal of sucrose, maltose and lactose in the measurement of dietary fiber. |
This product has been discontinued.
High purity Sucrase (maltase; Yeast) / β-Galactosidase (Aspergillus niger) for use in research, biochemical enzyme assays and analytical testing applications.
For other enzymes, view Carbohydrate Active enZYmes list.
Production and prebiotic properties of oligofructans from sugarcane juice fermentation by Bacillus subtilis TISTR 001.
Ninchan, B. & Noidee, C. (2021). 3 Biotech, 11(5), 1-11.
Oligofructans are potential biological substances that due to their distinctive properties have a positive health-promoting effect. This research aimed to produce oligofructans using sugarcane juice fermentation with Bacillus subtilis TISTR 001 and to study the prebiotic properties in vitro. The results showed that the maximum total oligofructans in the form of free fructose was 2.57% (w/v) at the 84th hour of fermentation with 0.17 g/g reducing sugar and a production yield of 0.031 g/L/h and maximum levansucrase activity of 1.57 × 106 U/mL at that time. The oligofructans contained in the fermented juice had potential functional ingredients that exhibited prebiotic properties that could resist the digestion of enzymes in the gastrointestinal tract under in vitro conditions with digestion of only 6.92%. In addition, the fermented juice promoted the growth of prebiotics, especially Bifidobacterium bifidum TISTR 2129 and inhibited the growth of pathogens using both single culturing and co-culturing with probiotics.
Hide AbstractEfficacy of reducing sugar and phenol–sulfuric acid assays for analysis of soluble carbohydrates in feedstuffs.
Hall, M. B. (2013). Animal Feed Science and Technology, 185(1), 94-100.
Reducing sugar (RSA) and phenol–sulfuric acid (PSA) assays are commonly used to analyze water-soluble carbohydrates. However, questions have arisen as to their accuracy for measurements of feedstuffs with diverse carbohydrate profiles. This study evaluated the efficacy of RSA and PSA as they would commonly be applied in feed analysis laboratories in measuring a variety of purified carbohydrates. Carbohydrates analyzed were glucose (Glc), fructose (Fru), galactose (Gal), sucrose (Suc), maltose (Mal), lactose (Lac), raffinose (Raf), and inulin (Inu). Variations on the methods used were PSA using Suc (PSA-Suc) or Glc (PSA-Glc) as standard sugars, and RSA with a 50:50 Glc:Fru blend as the standard with four hydrolysis methods: acid hydrolysis with 0.037 M sulfuric acid (RSA-H2SO4) or 0.5M hydrochloric acid (RSA-HCl), or enzymatic hydrolysis with invertase (RSA-Inv) or an enzyme blend including sucrase, α-glucosidase, and β-galactosidase (RSA-EnzBl). Recovery of carbohydrate was calculated on a dry matter (DM) basis as (carbohydrate detected g/kg DM)/(carbohydrate present kg/kg DM), with ‘close to’ complete recovery defined as values falling within the range of 920–1080 g/kg. Monosaccharide recovery did not differ between unhydrolyzed vs. hydrolyzed samples in RSA indicating no destruction of carbohydrate by hydrolysis method. For RSA, recoveries of Glc, Fru, and Gal were 979, 1042, and 706 g/kg, respectively. Such response differences among monosaccharides are inherent to RSA, and can affect carbohydrate recovery values. Methods that provided close to complete recovery by carbohydrate were: PSA-Suc and all RSA for Suc; PSA-Glc and RSA-EnzBl for Mal and Lac; PSA-Suc, RSA-H2SO4, RSA-HCl, and RSA-Inv for Raf; and RSA-H2SO4 and RS-HCl for Inu. None of the assays gave complete recovery of the diverse set of purified carbohydrates. Allowing a range of 920–1080 g/kg for recoveries on individual carbohydrates, RSA-H2SO4 and RSA-HCl would give the closest to complete recovery values for feeds such as forage and soybean in which Suc, Raf, and Inu were important, whereas RSA-EnzBl would be useful in feeds such as forages or dairy products when Suc, Mal, and Lac are of interest. The allowed 920–1080 g/kg range of acceptable recoveries addresses the point that given very diverse carbohydrate complements of feeds, these assays will not be extremely precise, but may still be serviceable for diet formulation. The most accurate measurements will be achieved by selection of detection method, hydrolysis method, and carbohydrate standard to give greatest recovery of predominant carbohydrates in feedstuffs.
Hide Abstract