| Content: | 50 assays per kit |
| Shipping Temperature: | Ambient |
| Storage Temperature: |
Short term stability: 2-8oC, Long term stability: See individual component labels |
| Stability: | > 1 year under recommended storage conditions |
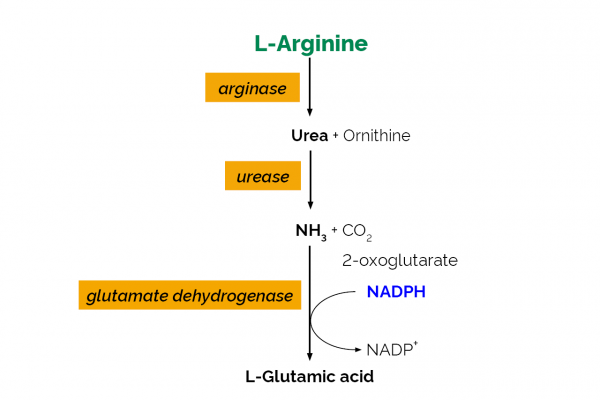
| Analyte: | Ammonia, L-Arginine, Nitrogen, Urea, YAN |
| Assay Format: | Spectrophotometer |
| Detection Method: | Absorbance |
| Wavelength (nm): | 340 |
| Signal Response: | Decrease |
| Linear Range: | 1.0 to 35 mg of L-arginine, or 0.2 to 7.0 μg of ammonia or 0.3 to 14 μg of urea per assay |
| Limit of Detection: |
0.07 mg/L (ammonia), 0.13 mg/L (urea), 0.37 mg/L (L-arginine) |
| Reaction Time (min): | ~ 20 min [ammonia (2 min), urea (6 min), L-arginine (7 min)] |
| Application examples: | Grape juice, wine must, wine and other materials (e.g. biological cultures, samples, etc.). |
| Method recognition: | Improved method |
The L-Arginine/Urea/Ammonia test kit is specific and a rapid measurement and analysis of L-arginine, urea and ammonia in grape juice/must and wine.
Note for Content: The number of manual tests per kit can be doubled if all volumes are halved. This can be readily accommodated using the MegaQuantTM Wave Spectrophotometer (D-MQWAVE).
Display all of our nitrogen assay kit products.

- Extended cofactors stability. Dissolved cofactors stable for > 1 year at 4oC.
- Improved assay format
- Very rapid reactions due to use of uninhibited glutamate dehydrogenase
- All enzymes supplied as stabilised suspensions
- Very competitive price (cost per test)
- All reagents stable for > 2 years after preparation
- Mega-Calc™ software tool is available from our website for hassle-free raw data processing
- Standard included
Megazyme “advanced” wine test kits general characteristics and validation.
Charnock, S. J., McCleary, B. V., Daverede, C. & Gallant, P. (2006). Reveue des Oenologues, 120, 1-5.
Many of the enzymatic test kits are official methods of prestigious organisations such as the Association of Official Analytical Chemicals (AOAC) and the American Association of Cereal Chemists (AACC) in response to the interest from oenologists. Megazyme decided to use its long history of enzymatic bio-analysis to make a significant contribution to the wine industry, by the development of a range of advanced enzymatic test kits. This task has now been successfully completed through the strategic and comprehensive process of identifying limitations of existing enzymatic bio-analysis test kits where they occurred, and then using advanced techniques, such as molecular biology (photo 1), to rapidly overcome them. Novel test kits have also been developed for analytes of emerging interest to the oenologist, such as yeast available nitrogen (YAN; see pages 2-3 of issue 117 article), or where previously enzymes were simply either not available, or were too expensive to employ, such as for D-mannitol analysis.
Hide AbstractGrape and wine analysis: Oenologists to exploit advanced test kits.
Charnock, S. C. & McCleary, B. V. (2005). Revue des Enology, 117, 1-5.
It is without doubt that testing plays a pivotal role throughout the whole of the vinification process. To produce the best possible quality wine and to minimise process problems such as “stuck” fermentation or troublesome infections, it is now recognised that if possible testing should begin prior to harvesting of the grapes and continue through to bottling. Traditional methods of wine analysis are often expensive, time consuming, require either elaborate equipment or specialist expertise and frequently lack accuracy. However, enzymatic bio-analysis enables the accurate measurement of the vast majority of analytes of interest to the wine maker, using just one piece of apparatus, the spectrophotometer (see previous issue No. 116 for a detailed technical review). Grape juice and wine are amenable to enzymatic testing as being liquids they are homogenous, easy to manipulate, and can generally be analysed without any sample preparation.
Hide AbstractDifferent Yeast Strain Effects on ‘King of the North’Wine Chemical, Chromatic, and Descriptive Sensory Characteristics.
Wang, Z., Svyantek, A., Kadium, V. R., Bogenrief, S. & Hatterman-Valenti, H. (2025). Fermentation, 11(5), 262.
‘King of the North’ (‘KON’), as a cold-hardy grape, has many advantages, such as tolerance to a wide range of soil conditions and harsh winter climate. Due to the adapting demand of North Dakota’s fruit and wine industry, optimized quality of wine from regionally productive grapevines is required. In this study, yeast strain, one of the primary fermentation tactics, was tested on ‘KON’ grapes. Five different commercial yeast strains, including 71B, EC1118, Maurivin B, Rhône 4600, and W15, were added to initiate fermentation. The analysis of grape must characteristics and the fermentation dynamic changes indicated a high correlation between color and acid metrics. Yeast strains have influenced the color dynamic changes and fermentation process. The panelist sensory evaluations confirmed that yeast strains contributed differently to the perceived aromas and flavors within ‘KON’ wines. Rose, apple, grape, and apricot aromas were distinguished in ‘KON’ wines. The lemon taste was the dominant flavor detected in ‘KON’ wines. However, wines were also varied based on the extent of the aroma or taste observed. Therefore, exploring the use of different yeast strains for fermentation provides information for further application to cold-hardy grape cultivars and other high-acid fruit, aiding winemakers in using North American grapes with diverse fruit chemistry.
Hide AbstractEffects of inoculation timing and mixed fermentation with Metschnikowia pulcherrima and Saccharomyces cerevisiae on the aroma and sensory properties of Falanghina wine.
Coppola, F., Testa, B., Cozzolino, R., Karaulli, J., Pannella, G., Di Renzo, M., Matarazzo, C., Succi, M. & Iorizzo, M. (2025). European Food Research and Technology, 1-19.
In recent years, non-Saccharomyces yeasts, including Metschnikowia pulcherrima, have gained renewed interest in biotechnology applied to the wine industry due to their pro-technological properties. M. pulcherrima has been particularly studied for its antimicrobial activity, its potential to produce wines with a low alcohol content, and its enzymatic activities that enhance some sensory characteristics of wines, especially when used in co-culture with Saccharomyces cerevisiae. This study evaluated the production of Falanghina wine using M. pulcherrima AS3C1 as an initial starter, followed by sequential inoculation with a commercial S. cerevisiae after 2 and 4 days. Moreover, a parallel vinification was performed using S. cerevisiae as a single starter culture. Volatile Organic Compounds (VOCs) data, obtained through Headspace Solid phase Microextraction couple to Gas Chromatography-Mass Spectrometry (HS-SPME/GC–MS), were analyzed using multivariate statistical methods (PCA and sPLS-DA). The results showed significant differences in the VOC profiles of the wines produced. Notably, the wine obtained through sequential inoculation of M. pulcherrima AS3C1 followed by S. cerevisiae after 4 days received the highest overall sensory evaluation by the panel test. This wine was distinguished by its elevated levels of ethyl acetate, phenylethyl acetate, benzeneethanol and 2-methylpropanol.
Hide AbstractClinically relevant mutations in regulatory regions of metabolic genes facilitate early adaptation to ciprofloxacin in Escherichia coli.
Pal, A., Ghosh, D., Thakur, P., Nagpal, P., Irulappan, M., Maruthan, K., Mukherjee, S., Patil, N. G., Dutta, T., Veeraraghavan, B. & Vivekanandan, P. (2024). Nucleic Acids Research, 52(17), 10385-10399.
The genomic landscape associated with early adaptation to ciprofloxacin is poorly understood. Although the interplay between core metabolism and antimicrobial resistance is being increasingly recognized, mutations in metabolic genes and their biological role remain elusive. Here, we exposed Escherichia coli to increasing gradients of ciprofloxacin with intermittent transfer-bottlenecking and identified mutations in three non-canonical targets linked to metabolism including a deletion (tRNA-ArgΔ414-bp) and point mutations in the regulatory regions of argI (ARG box) and narU. Our findings suggest that these mutations modulate arginine and carbohydrate metabolism, facilitate anaerobiosis and increased ATP production during ciprofloxacin stress. Furthermore, mutations in the regulatory regions of argI and narU were detected in over 70% of sequences from clinical E. coli isolates and were overrepresented among ciprofloxacin-resistant isolates. In sum, we have identified clinically relevant mutations in the regulatory regions of metabolic genes as a central theme that drives physiological changes necessary for adaptation to ciprofloxacin stress.
Hide AbstractChemical Characterization of Cider Produced in Hardanger-From Juice to Finished Cider.
Ovsthus, I., Martelanc, M., Albreht, A., Radovanović Vukajlović, T., Česnik, U. & Mozetič Vodopivec, B. (2024). Beverages, 10(3), 73.
Our investigation delves into the previously uncharted territory of cider composition from Norway. This study aimed to obtain an overview of the qualitative and quantitative compositions of general chemical parameters, polyphenols (individual and total expressed as gallic acids equivalents), selected esters, and selected C6-alcohols in ciders with the PDO label Cider from Hardanger. In total, 45 juice and cider samples from the fermentation process were collected from 10 cider producers in Hardanger in 2019, 2020, and 2021. Individual sugars, acids, ethanol, and 13 individual phenols were quantified using HPLC-UV/RI. Seven ethyl esters of fatty acids, four ethyl esters of branched fatty acids, ten acetate esters, two ethyl esters of hydroxycinnamic acids, and four C6-alcohols were quantified using HS-SPME-GC-MS. For samples of single cultivars (‘Aroma’, ‘Discovery’, ‘Gravenstein’, and ‘Summerred’), the sum of the measured individual polyphenols in the samples ranges, on average, from 79 to 289 mg L−1 (the lowest for ‘Summerred’ and highest for ‘Discovery’ and ‘Gravenstein’). Chlorogenic acid was the most abundant polyphenol in all samples. Ethyl butyrate, ethyl hexanoate, ethyl octanoate, ethyl decanoate, ethyl isobutyrate, ethyl 2-methylbutyrate, isoamyl acetate, and hexanol were present at concentrations above the odour threshold and contributed to the fruity flavour of the Cider from Hardanger.
Hide AbstractExploring the influence of grape tissues on the concentration of wine volatile compounds.
Blackford, C. L., Trengove, R. D. & Boss, P. K. (2021). Australian Journal of Grape and Wine Research, In Press.
Background and Aims: Knowledge of varietal wine flavour and aroma compounds has improved, but gaps exist concerning how grape composition impacts wine style. This work aimed to explore the influence that different grape tissues can have on the volatile profiles of wines. Methods and Results: Riesling and Cabernet Sauvignon berries were separated into skin, flesh and seeds. Two sets of fermentations were performed using separated tissues: one using an equal mass of each tissue and another where the amount of each tissue in 25 g of berries was fermented. When an equal mass of tissue was used, the seed-derived wines had a higher concentration of esters than that produced from other grape tissues. Those produced using skins had the highest concentration of lipoxygenase pathway-derived compounds, and, for Riesling, a higher concentration of monoterpenes. When the proportional amounts of each tissue found per berry were used, the flesh-derived wines generally had a higher concentration of many wine volatiles compared to the other tissues. This reflects the greater proportion of flesh tissue in the berry compared to skin and seeds. Conclusions: Seed-derived compounds can enhance ester biosynthesis during fermentation and skins appear to have high lipoxygenase pathway activity. Nevertheless, the flesh makes up such a large proportion of the whole berry that it has the major influence on volatile profiles of whole berry fermentations. Significance of the Study: Different berry tissues can alter wine composition in unique ways, and this can inform strategies to alter wine composition through vineyard management or the selection of new germplasm.
Hide AbstractArginase activity characterization during alcoholic fermentation by sequential inoculation with non-Saccharomyces and Saccharomyces Yeast.
Benucci, I. & Esti, M. (2021). Food and Bioprocess Technology, 1-8.
Arginine uptake and yeast arginase activity were studied throughout the alcoholic fermentation of a white grape must (Vitis vinifera L. cv Fiano), carried out by sequential inoculation (Torulaspora delbrueckii and S. cerevisiae, TD-SC) and compared with a S. cerevisiae single fermentation (SC). In both samples, yeast assimilable nitrogen (YAN) consumption was mainly during the early phase of alcoholic fermentation (before S. cerevisiae addition in the sequential fermentation). T. delbrueckii alone and S. cerevisiae similarly metabolized YAN, which was further consumed in TD-SC sample following the S. cerevisiae inoculation. The only relevant arginine uptake was found about 3 days after the first inoculum (simultaneously with the greatest YAN consumption) and it appeared to be higher in SC (0.24 g/L) than in TD-SC (0.12 g/L). The kinetic parameters, estimated by means of the Hill equation, and in particular Vmax values (56.0 ± 1.6 U/mgBSAeq at 48 h for SC and 73.3 ± 2.7 U/mgBSAeq at 66 h for TD-SC) indicated the maximum arginase activity at the same point of time corresponding to the lowest YAN amount (48 h for S. cerevisiae and 66 h for T. delbrueckii).
Hide AbstractAroma and Sensory Profiles of Sauvignon Blanc Wines from Commercially Produced Free Run and Pressed Juices.
Parish-Virtue, K., Herbst-Johnstone, M., Bouda, F., Fedrizzi, B., Deed, R. C. & Kilmartin, P. A. (2021). Beverages, 7(2), 29.
Sauvignon blanc is the most important grape cultivar within the New Zealand wine industry, and wines from the Marlborough region are renowned for their intense aromas including tropical, passionfruit, and green capsicum. Quality Sauvignon blanc wines are usually made from free run juice, although press fractions can be included. The chemical aroma composition and sensory profiles of two wine sets made from three press fractions (free run, light press and heavy press) were compared. The compounds 3-mercaptohexan-1-ol and 3-mercaptohexyl acetate were found to decrease between free run and heavily pressed wines while hexyl acetate, hexanol, and benzyl alcohol increased. The accompanying sensory analysis showed that free run wines were marked by aromas of Passionfruit/sweaty, Boxwood and Fresh green capsicum, while the heavy pressed wines were described by French vanilla/bourbon, Floral and Banana lolly attributes, consistent with the aroma chemical composition.
Hide AbstractNanocurcumin and arginine entrapped injectable chitosan hydrogel for restoration of hypoxia induced endothelial dysfunction.
Mohandas, A. & Rangasamy, J. (2020). International Journal of Biological Macromolecules, 166, 471-482.
Hypoxia is a condition that gradually leads to ischemic damages in organs which is marked by poor tissue perfusion. Depending on the severity of the condition, revascularisation therapies are needed for reducing the risk of organ dysfunction. This study was aimed at developing an injectable nanocurcumin and arginine incorporated chitosan hydrogel (nC/R) that can prevent hypoxia induced endothelial damage. The prepared hydrogel has shear thinning, stable and injectable nature. The (nC and nC/R) hydrogels showed significant antioxidant activity and biodegradation in vitro. The release of curucmin and arginine from the nC/R was found to be higher at acidic pH, which predominates in an ischemic site. To mimic low oxygen environment, an in vitro hypoxic endothelial dysfunction model was developed which showed decreased expressions of phosphorylated eNOS (serine 1177) when compared to the cells cultured in normoxic condition. In vitro tube formation assay demonstrated the protective effect of nC/R towards hypoxia induced reduction of tube width. The nC/R hydrogel was found to enhance phosphorylation of eNOS at serine 1177 site in cultured endothelial cells subjected to hypoxia. Therefore, nC/R hydrogel could effectively deliver both curcumin and arginine and therapeutically reduce the effect of hypoxia induced endothelial dysfunction.
Hide AbstractFunctional Applications of Polyarginine-Hyaluronic Acid-Based Electrostatic Complexes.
Kale, N. R., Dutta, D., Carstens, W., Mallik, S. & Quadir, M. (2020). Bioelectricity, 2(2), 158-166.
Background: Electrostatic complexes of poly (l-Arginine) (pArg) and hyaluronic acid (HA) have been investigated for their functional applications to supply free or polymeric form of l-Arginine (Arg) to target cells. As a vital amino acid, Arg plays significant role in multitude of pathophysiological processes ranging from wound healing to cancer. However, serum arginase expression and toxicity of Arg at cellular level renders exogenous delivery of this amino acid a challenging task. We showed that polyarginine-hyaluronic acid ionic nanocomplexes (pArg-HA iNCs) could be an effective way to deliver Arg to target cell populations. Materials and Methods: These electrostatic complexes were prepared by mixing HA (average m.w. of 200 kDa) with pArg (m.w. 5-15 kDa; Sigma) in aqueous solutions and purifying over glycerol. Nanocomplexes were characterized for their particle size, surface charge, capacity to release l-Arg, and intracellular uptake of complexes. Results: Synthesized nanocomplexes showed hydrodynamic diameter ranging from 140-306 nm depending on the content of pArg or HA within the formulation. With surface charge (ζ-potential) of -29 mV, the nanocomplexes showed pH-dependent release of Arg. At pH 7.4, pArg-HA iNCs released 30% of the total Arg-content, while at pH 5.0, 60% of Arg was released after 24 h. These electrostatically stabilized complexes were found to promote growth of human dermal fibroblasts (HDF) in wound-healing assay and increased nitric oxide (NO) activity in these cells in a time-dependent manner. Nanocomplexes also showed cellular uptake and enhanced dose-dependent toxicity against two pancreatic cancer cell lines, i.e. MIA PaCa-2 and Panc-1. Interestingly, the cytotoxic effect was synergized upon pre-treatment of the cells with a frontline chemotherapeutic agent, gemcitabine (GEM), and was not observed when the cells were treated with Arg alone. Conclusion: As such, this communication shows the prospect of pArg-HA iNC electrostatic nanocomplexes to interact and interfere with intracellular Arg metabolic machinery conducive to rescuing different pathological conditions.
Hide AbstractParish-Virtue, K., Herbst-Johnstone, M., Bouda, F. & Fedrizzi, B. (2019). Food Chemistry, 271, 747-752.
Sauvignon blanc grapes were exposed to an ultra-violet (UV) light source post-hand harvest (whole bunches) or post-machine harvest. The thiol precursors S-3-(hexan-1-ol)-L-cysteine (Cys-3MH) and S-3-(hexan-1-ol)-L-glutathione (GSH-3MH) were quantified in the juices before and after UV treatment. Results showed that irradiation of the grapes with UV light had little to no effect on the thiol precursors. Wines were fermented from the corresponding juices and 18 aroma compounds were quantified. Differences were found between UV treatments of the wines for 3-mercaptohexanol, hexan-1-ol, ethyl butanoate, ethyl hexanoate, ethyl octanoate and phenylethyl alcohol. However, these changes were not significant (p < 0.05) for both grape media trialled. Future studies involving larger sample sizes and replicate numbers should be completed in order to ascertain any changes in aroma chemistry as a result of UV light application to grapes postharvest.
Hide AbstractEnglezos, V., Rantsiou, K., Cravero, F., Torchio, F., Giacosa, S., Ortiz-Julien, A., Gerbi, V., Rolle, L. & Cocolin, L. (2018). Food Research International, 109, 298-309.
The use of mixed fermentations with Starmerella bacillaris and Saccharomyces cerevisiae is gaining attention in recent years due to their ability to modulate the metabolites production of enological interest. In the present study, four of the most popular planted red grape varieties (Cabernet sauvignon, Merlot, Pinot noir and Shiraz) were fermented using the aforementioned species and two different inoculation protocols (inoculation of S. cerevisiae after 24 and 48 h from the Starm. bacillaris inoculation), in order to evaluate their impact on the volatile composition and chromatic characteristics of wines. Analysis from chemical composition showed that titratable acidity and glycerol content exhibited marked differences among wines after fermentation. For volatile compounds, mixed fermented wines using an inoculation delay of 48 h led to reduction of volatile compounds (mainly esters). A shorter 24 h delay produced wines with higher values of color intensity than pure fermented wines. The differences observed between the inoculation protocols can be explained by the growth dynamics of both species during fermentation. These findings suggest that mixed fermentations posed a great potential in reducing metabolites which are considered negative for wine quality (mainly ethyl acetate and volatile fatty acids) and with an improvement of the chromatic profile of the wines.
Hide AbstractBenucci, I., Fiorelli, V., Lombardelli, C., Liburdi, K. & Esti, M. (2017). LWT-Food Science and Technology, 82, 268-273.
The kinetic characterization of arginase activity of a commercial S. cerevisiae strain was carried out for the first time, estimating the kinetic parameters (Vmax, K0.5 and Vmax/K0.5) throughout alcoholic fermentation in order to investigate the catalytic efficiency of the enzyme and its ability in metabolizing arginine to sustain biosynthetic processes. Alcoholic fermentation was carried out at three different temperatures (15, 20, 25°C) in semi-synthetic grape juice added with arginine at usual maximal concentration (1 g L-1) in grape must. Arginine uptake was quite constant throughout fermentation process and it was more effectively assimilated during high temperature fermentation (20 and 25°C) than at 15°C. The sigmoidal behavior of yeast arginase kinetic curves, well fitted to the Hill equation, indicated a mechanism of positive cooperativity for the trimeric enzyme. The highest Vmax (4740.0 U mg-1BSAeq) and the maximal catalytic efficiency (78.87 min-1) were observed when fermentation was at 20°C approximately 3 days after the inoculum. Moreover, the K0.5 value was similar (53–60 mg mL-1) when maximal catalytic efficiency was achieved, thus indicating that the affinity of enzyme for the substrate is not altered by fermentation temperature which only affected product release velocity and therefore Vmax/K0.5 ratio.
Hide AbstractWang, C., Esteve-Zarzoso, B., Cocolin, L., Mas, A. & Rantsiou, K. (2015). Food Research International, 78, 195-200.
The present study analyzed the viable and/or culturable populations of Saccharomyces cerevisiae, Hanseniaspora uvarum and Starmerella bacillaris (synonym Candida zemplinina) during laboratory grape must fermentation, in order to investigate the interaction between the three species considered. Firstly, population dynamics during wine fermentation were followed by culture-dependent techniques, and non-Saccharomyces yeast became non-culturable at late stages of fermentation when S. cerevisiae dominated. Four different culture-independent techniques were further applied to detect viable yeast cells at the late stage of fermentation. Both quantitative PCR techniques applied, namely ethidium monoazide bromide (EMA)-qPCR and Reverse Transcription (RT)-qPCR, detected H. uvarum and Starm. bacillaris at a concentration of 105 to 106 cells/mL. These non-culturable cells had membranes impermeable to EMA and stable rRNA. The background signals from dead cells did not interfere with the quantification of viable cells in wine samples by EMA-qPCR technique. As a qualitative culture-independent technique, DGGE technique was coupled with EMA treatment (EMA-PCR-DGGE) or with RT (RT-PCR-DGGE). With EMA-PCR-DGGE non-Saccharomyces species during fermentation were detected although it was limited by the predominance of S. cerevisiae.
Hide Abstract