| Content: | 1,000 Units |
| Shipping Temperature: | Ambient |
| Storage Temperature: | 2-8oC |
| Formulation: | In 3.2 M ammonium sulphate (stabilised with BSA) |
| Physical Form: | Suspension |
| Stability: | > 4 years at 4oC |
| Enzyme Activity: | α-Galactosidase |
| EC Number: | 3.2.1.22 |
| CAZy Family: | GH27 |
| CAS Number: | 9025-35-8 |
| Synonyms: | alpha-galactosidase; alpha-D-galactoside galactohydrolase |
| Source: | Guar |
| Molecular Weight: | 44,500 |
| Concentration: | Supplied at ~ 500 U/mL |
| Expression: | Purified from Guar seed |
| Specificity: | Hydrolysis of terminal, non-reducing α-D-galactose residues in α-D-galactosides, including galactose oligosaccharides, galactomannans and galactolipids. |
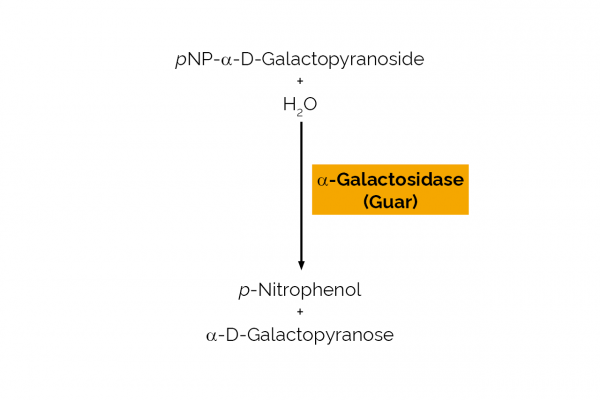
| Specific Activity: | ~ 50 U/mg (40oC, pH 4.5 on p-nitrophenyl-α-D-galactopyranoside, before addition of BSA) |
| Unit Definition: | One Unit of activity is the amount of enzyme required to release one µmole of p-nitrophenol (pNP) per minute from p-nitrophenyl-α-D-galactopyranoside per min at pH 4.5 and 40oC. |
| Temperature Optima: | 40oC |
| pH Optima: | 4.5 |
| Application examples: | Applications in carbohydrate and glycobiology research. |
This product has been discontinued (read more).
High purity α-Galactosidase (Guar) for use in research, biochemical enzyme assays and in vitro diagnostic analysis.
Show our full range of CAZy and glycobiology enzyme products.
McCleary, B. V. (1994). “Methods in Carbohydrate Chemistry”, Vol. X, (J. N. BeMiller, D. J. Manners and R. J. Sturgeon, Eds.), John Wiley & Sons Inc., pp. 175-182.
A number of methods have been described for the analysis of the fine structure of galactomannans, i.e., the distribution of D-galactosyl units along the D-mannan backbone (1). Such studies include the analysis of x-ray diffraction data of stretched fibers of galactomannans (2,3), 1H- and 13C-nmr (nuclear magnetic resonance) of native and partially depolymerized galacto¬mannans (4) and a range of chemical procedures (5-7), including those employing a detailed theoretical analysis of the kinetics of reaction (8). An alternative approach involves the characterization and quantification of the oligosaccharides produced on hydrolysis of galactomannans by highly purified and well-characterized β-mannanases (EC 3.2.1.78) (9,10). The β-mannanases employed were purified to homogeneity by affinity chromatography on gIucornannan-AH-Sepharose 4B. They were characterized by a range of physicochemicai procedures by determining the kinetics of their action on β-mannooligosaccharides, and by characterizing the structures of oligosaccharides produced on hydrolysis of galactomannans and glucomannans (11). From these studies, a basic model describing the subsite binding requirements of all the β-mannanases examined was proposed (Fig. 1). This model was then modified to account for the slight differences noted in the types of oligosaccharides produced by β-mannanases from different sources. The β-mannanases which differ most significantly in their action patterns on galactomannans are those from Aspergillus niger culture filtrates and from germinated guar seed.
Hide AbstractMcCleary, B. V. (1988). “Methods in Enzymology”, Volume 160, (H. Gilbert, Ed.), Elsevier Inc., pp. 627-632.
α-Galactosidase has been shown to occur in a wide range of plants and animals and to be synthesized by microorganisms. This enzyme has been purified from several sources using conventional chromatographic procedures and a range of affinity supports. Of the affinity procedures, that employing N-ɛ-aminocaproyl-α-D-galactopyranosylamine coupled to Sepharose 4B as described by Harpaz et al. is effective and reliable and can be used to purify α-galactosidase from a wide range of biological materials. The affinity technique described by Harpaz et al. was employed to purify α-galactosidase from green coffee beans and from soybean seed. However, neither of these materials is a good source of this activity. This chapter describes the large-scale purification of α-galactosidases with high activity on galactomannan from germinated seeds of lucerne and guar employing the affinity matrix.
Hide AbstractMallett, I., McCleary, B. V. & Matheson, N. K. (1987). Phytochemistry, 26(7), 1889-1894.
Galactomannan has been extracted from the endosperm of seeds of Gleditsia triacanthos (honey locust) at different stages of development, when the seed was accumulating storage material. Properties of the different samples have been studied. The molecular size distribution became more disperse as galactomannan accumulated and the galactose: mannose ratio decreased slightly. Some possible reasons for these changes are discussed.
Hide AbstractDea, I. C. M., Clark, A. H. & McCleary, B. V. (1986). Carbohydrate Research, 147(2), 275-294.
A range of galactomannans varying widely in the contents of D-galactose have been compared for self-association and their interaction properties with agarose and xanthan. Whereas, in general, the most interactive galactomannans are those in which the (1→4)-β-D-mannan chain is least substituted by α-D-galactosyl stubs, evidence is presented which indicates that the distribution of D-galactosyl groups along the backbone (fine structure) can have a significant effect on the interaction properties. For galactomannans containing <30% of D-galactose, those which contain a higher frequency of unsubstituted blocks of intermediate length in the β-D-mannan chain are most interactive. For galactomannans containing >40% of D-galactose, those which contain a higher frequency of exactly alternating regions in the β-D-mannan chain are most interactive. This selectivity, on the basis of galactomannan fine-structure, in mixed polysaccharide interactions in vitro could mimic the selectivity of binding of branched plant-cell-wall polysaccharides in biological systems.
Hide AbstractEffect of the molecular fine structure of galactomannans on their interaction properties - the role of unsubstituted sides.
Dea, I. C. M., Clark, A. H. & McCleary, B. V. (1986). Food Hydrocolloids, 1(2), 129-140.
A range of galactomannans varying widely in the content of D-galactose have been compared for self-association, and their interaction properties with agarose and xanthan. The results presented indicate that in general the most interactive galactomannans are those in which the D-mannan main chain bears fewest D-galactose stubs, and confirm that the distribution of D-galactose groups along the main chain can have a significant effect on the interactive properties of the galactomannans. It has been shown that freeze — thaw precipitation of galactomannans requires regions of totally unsubstituted D-mannose residues along the main chain, and that a threshold for significant freeze — thaw precipitation occurs at a weight-average length of totally unsubstituted residues of approximately six. For galactomannans having structures above this threshold their interactive properties with other polysaccharides are controlled by structural features associated with totally unsubstituted regions of the D-mannan backbone. In contrast, for galactomannans below this threshold, their interactive properties are controlled by structural features associated with unsubstituted sides of D-mannan backbone.
McCleary, B. V., Clark, A. H., Dea, I. C. M. & Rees, D. A. (1985). Carbohydrate Research, 139, 237-260.
The distribution of D-galactosyl groups along the D-mannan backbone (fine structure) of carob and guar galactomannans has been studied by a computer analysis of the amounts and structures of oligosaccharides released on hydrolysis of the polymers with two highly purified β-D-mannanases isolated from germinated guar seed and from Aspergillus niger cultures. Computer programmes were developed which accounted for the specific subsite-binding requirements of the β-D-mannanases and which simulated the synthesis of galactomannan by processes in which the D-galactosyl groups were transferred to the growing D-mannan chain in either a statistically random manner or as influenced by nearest-neighbour/second-nearest-neighbour substitution. Such a model was chosen as it is consistent with the known pattern of synthesis of similar polysaccharides, for example, xyloglucan; also, addition to a preformed mannan chain would be unlikely, due to the insoluble nature of such polymers. The D-galactose distribution in carob galactomannan and in the hot- and cold-water-soluble fractions of carob galactomannan has been shown to be non-regular, with a high proportion of substituted couplets, lesser amounts of triplets, and an absence of blocks of substitution. The probability of sequences in which alternate D-mannosyl residues are substituted is low. The probability distribution of block sizes for unsubstituted D-mannosyl residues indicates that there is a higher proportion of blocks of intermediate size than would be present in a galactomannan with a statistically random D-galactose distribution. Based on the almost identical patterns of amounts of oligosaccharides produced on hydrolysis with β-D-mannanase, it appears that galactomannans from seed of a wide range of carob varities have the same fine-structure. The D-galactose distribution in guar-seed galactomannan also appears to be non-regular, and galactomannans from different guar-seed varieties appear to have the same fine-structure.
Hide AbstractEnzymes metabolizing polysaccharides and their application to the analysis of structure and function of glycans.
Matheson, N. K. & McCleary, B. V. (1985). “The Polysaccharides”, Volume 3, (G. O. Aspinall, Ed.), Academic Press Inc., pp. 1-105.
Enzymes metabolizing polysaccharides were used in such processes as baking and brewing for countless centuries before the relationship between the chemical structure of the polysaccharides and their modification by enzymes was known. In the past two centuries, studies of the structures and functions of polysaccharides involved in the storage of chemical energy, in the structural parts of tissues, and as information carriers, as well as the enzymes that metabolize them, have been carried out.
Hide AbstractMcCleary, B. V. (1983). Carbohydrate Research, 111(2), 297-310.
β-D-Mannosidase (β-D-mannoside mannohydrolase EC 3.2.1.25) was purified 160-fold from crude gut-solution of Helix pomatia by three chromatographic steps and then gave a single protein band (mol. wt. 94,000) on SDS-gel electrophoresis, and three protein bands (of almost identical isoelectric points) on thin-layer iso-electric focusing. Each of these protein bands had enzyme activity. The specific activity of the purified enzyme on p-nitrophenyl β-D-mannopyranoside was 1694 nkat/mg at 40° and it was devoid of α-D-mannosidase, β-D-galactosidase, 2-acet-amido-2-deoxy-D-glucosidase, (1→4)-β-D-mannanase, and (1→4)-β-D-glucanase activities, almost devoid of α-D-galactosidase activity, and contaminated with <0.02% of β-D-glucosidase activity. The purified enzyme had the same Km for borohydride-reduced β-D-manno-oligosaccharides of d.p. 3-5 (12.5mM). The initial rate of hydrolysis of (1→4)-linked β-D-manno-oligosaccharides of d.p. 2-5 and of reduced β-D-manno-oligosaccharides of d.p. 3-5 was the same, and o-nitrophenyl, methylumbelliferyl, and naphthyl β-D-mannopyranosides were readily hydrolysed. β-D-Mannobiose was hydrolysed at a rate ~25 times that of 61-α-D-galactosyl-β-D-mannobiose and 63-α-D-galactosyl-β-D-mannotetraose, and at ~90 times the rate for β-D-mannobi-itol.
Hide AbstractMcCleary, B. V. (1983). Phytochemistry, 22(3), 649-658.
Hydrolysis of galactomannan in endosperms of germinating guar is due to the combined action of three enzymes, α-galactosidase, β-mannanase and exo-β-mannanase. α-Galactosidase and exo-β-mannanase activities occur both in endosperm and cotyledon tissue but β-mannanase occurs only in endosperms. On seed germination, β-mannanase and endospermic α-galactosidase are synthesized and activity changes parallel galactomannan degradation. Galactomannan degradation and synthesis of these two enzymes are inhibited by cycloheximide. In contrast, endospermic exo-β-mannanase is not synthesized on seed germination, but rather is already present throughout endosperm tissue. It has no action on native galactomannan. α-Galactosidase, β-mannanase and exo-β-mannanase have been purified to homogeneity and their separate and combined action in the hydrolysis of galactomannan and effect on the rate of uptake of carbohydrate by cotyledons, studied. Results obtained indicated that these three activities are sufficient to account for galactomannan degradation in vivo and, further, that all three are required. Cotyledons contain an active exo-β-mannanase and sugar-uptake experiments have shown that cotyledons can absorb mannobiose intact, indicating that this enzyme is involved in the complete degradation of galactomannan on seed germination.
Hide AbstractMcCleary, B. V., Nurthen, E., Taravel, F. R. & Joseleau, J. P. (1983). Carbohydrate Research, 118, 91-109.
Treatment of hot-water-soluble carob galactomannan with β-D-mannanases from A. niger or lucerne seed affords an array of D-galactose-containing β-D-mannosaccharides as well as β-D-manno-biose, -triose, and -tetraose (lucerne-seed enzyme only). The D-galactose-containing β-D-mannosaccharides of d.p. 3–9 produced by A. niger β-D-mannanase have been characterised, using enzymic, n.m.r., and chemical techniques, as 61-α-D-galactosyl-β-D-mannobiose, 61-α-D-galactosyl-β-D-mannotriose, 63,64-di-α-D-galactosyl-β-D-mannopentaose (the only heptasaccharide), and 63,64-di-α-D-galactosyl-β-D-mannohexaose, 64,65-di-α-D-galactosyl-β-D-mannohexaose, and 61, 63,64-tri-α-D-galactosyl-β-D-mannopentaose (the only octasaccharides). Four nonasaccharides have also been characterised. Penta- and hexa-saccharides were absent. Lucerne-seed β-D-mannanase produced the same branched tri-, tetra- and hepta-saccharides, and also penta- and hexa-saccharides that were characterised as 61-α-D-galactosyl-β-D-mannotetraose, 63-α-D-galactosyl-β-D-mannotetraose, 61,63-di-α-D-galactosyl-β-D-mannotetraose, 63-α-D-galactosyl-β-D-mannopentaose, and 64-α-D-galactosyl-β-D-mannopentaose. None of the oligosaccharides contained a D-galactose stub on the terminal D-mannosyl group nor were they substituted on the second D-mannosyl residue from the reducing terminal.
Hide AbstractMcCleary, B. V. & Matheson, N. K. (1983). Carbohydrate Research, 119, 191-219.
Purified (1→4)-β-D-mannanase from Aspergillus niger and lucerne seeds has been incubated with mannosaccharides and end-reduced (1→4)-β-D-mannosaccharides and, from the products of hydrolysis, a cyclic reaction-sequence has been proposed. From the heterosaccharides released by hydrolysis of the hot-water-soluble fraction of carob galactomannan by A. niger β-D-mannanase, a pattern of binding between the β-D-mannan chain and the enzyme has been deduced. The products of hydrolysis with the β-D-mannanases from Irpex lacteus, Helix pomatia, Bacillus subtilis, and lucerne and guar seeds have also been determined, and the differences from the action of A. niger β-D-mannanase related to minor differences in substrate binding. The products of hydrolysis of glucomannan are consistent with those expected from the binding pattern proposed from the hydrolysis of galactomannan.
Hide AbstractMcCleary, B. V. (1982), Carbohydrate Research, 101(1), 75-92.
A β-D-mannoside mannohydrolase enzyme has been purified to homogeneity from germinated guar-seeds. Difficulties associated with the extraction and purification appeared to be due to an interaction of the enzyme with other protein material. The purified enzyme hydrolysed various natural and synthetic substrates, including β-D-manno-oligosaccharides and reduced β-D-manno-oligosaccharides of degree of polymerisation 2 to 6, as well as p-nitrophenyl, naphthyl, and methylumbelliferyl β-D-mannopyranosides. The preferred, natural substrate was β-D-mannopentaose, which was hydrolysed at twice the rate of β-D-mannotetraose and five times the rate of β-D-mannotriose. This result, together with the observation that α-D-mannose is released on hydrolysis, indicates that the enzyme is an exo-β-D-mannanase.
Hide AbstractMcCleary, B. V., Taravel, F. R. & Cheetham, N. W. H. (1982). Carbohydrate Research, 104(2), 285-297.
N.m.r., enzymic, and chemical techniques have been used to characterise the D-galactose-containing tri- and tetra-saccharides produced on hydrolysis of carob and L. leucocephala D-galacto-D-mannans by Driselase β-D-mannanase. These oligosaccharides were shown to be exclusively 61-α-D-galactosyl-β-D-mannobiose and 61-α-D-galactosyl-β-D-mannotriose. Furthermore, these were the only D-galactose-containing tri- and tetra-saccharides produced on hydrolysis of carob D-galacto-D-mannan by β-D-mannanases from other sources, including Bacillus subtilis, Aspergillus niger, Helix pomatia gut solution, and germinated legumes. Acid hydrolysis of lucerne galactomannan yielded 61-α-D-galactosyl-β-D-mannobiose and 62-α-D-galactosyl-β-D-mannobiose.
Hide AbstractMcCleary, B. V., Amado, R., Waibel, R. & Neukom, H. (1981). Carbohydrate Research, 92(2), 269-285.
Guar galactomannan has been modified by treatment with an α-D-galactosidase A preparation from lucerne seeds. This enzyme was purified by affinity chromatography on N-ϵ-aminocaproyl-α-D-galactopyranosylamine linked to Sepharose 4B, had a high activity towards galactomannans, and was completely devoid of β-D-mannanase. On incubation for 2 h, this enzyme removed > 75% of the galactose from guar galactomannan with no concurrent decrease in viscosity. Eventual decrease in viscosity was associated with the formation of insoluble, mannan-type precipitates. This phenomenon, although directly related to the galactose content of the galactomannan, was also time-dependent. The limiting viscosity numbers calculated for the “mannan backbones” of α-D-galactosidase-treated, guar galactomannan having galactose-mannose ratios of 38:62 to 15:85 were the same. Modified, guar galactomannan (at 0.4% w/v) having a galactose-mannose ratio of 20:80, or less, forms a gel on storage at 4° over several weeks. Also, gel particles form when solutions of these galactomannans are passed through a freeze-thaw cycle. Samples containing < 10% of galactose rapidly precipitate from solution even at 30°. The interaction of guar galactomannan with xanthan is greatly increased by removal of galactose residues. Samples having galactose-mannose ratios of ~19:81 interact with xanthan to essentially the same degree as carob galactomannan (Gal/Man = 23:77).
Hide AbstractAn enzymic technique for the quantitation of galactomannan in guar Seeds.
McCleary, B. V. (1981). Lebensmittel-Wissenschaft & Technologie, 14, 56-59.
An enzymic technique has been developed for the rapid and accurate quantitation of the galactomannan content of guar seeds and milling fractions. The technique involves the measurement of the galactose component of galactomannans using galactose dehydrogenase. The galactomannans are converted to galactose and manno-oligosaccharides using partially purified enzymes from a commercial preparation and from germinated guar seeds. Simple procedures have been devised for the preparation of these enzymes. Application of the technique to a number of guar varieties gave values for the galactomannan content ranging from 22.7 to 30.8% of seed weight.
Hide AbstractMcCleary, B. V. (1980). “Mechanisms of Saccharide Polymerization and Depolymerization”, (J. John. Marshall, Ed.), Academic Press Inc., pp. 285-300.
D-Galacto-D-mannans occur in the endosperms of a wide range of leguminous seeds in amounts varying from 0.1% (soybean) to 45% (Cassia brewsterii) of seed weight (1). The polysaccharides from different species have different proportions of D-galactose and D-mannose, but essentially always consist of a β-1,4-linked mannan backbone with single D-galactose branches lined α-1,6 (2).
Hide AbstractMcCleary, B. V. (1979). Phytochemistry, 18(5), 757-763.
β-Mannanase activities in the commercial enzyme preparations Driselase and Cellulase, in culture solutions of Bacillus subtilis (TX1), in commercial snail gut (Helix pomatia) preparations and in germinated seeds of lucerne, Leucaena leucocephala and honey locust, have been purified by substrate affinity chromatography on glucomannan-AH-Sepharose. On isoelectric focusing, multiple protein bands were found, all of which had β-mannanase activity. Each preparation appeared as a single major band on SDS-polyacrylamide gel electrophoresis. The enzymes varied in their final specific activities, Km values, optimal pH, isoelectric points and pH and temperature stabilities but had similar MWs. The enzymes have different abilities to hydrolyse galactomannans which are highly substituted with galactose. The preparations Driselase and Cellulase contain β-mannanases which can attack highly substituted galactomannans at points of single unsubstituted D-mannosyl residues if the D-galactose residues in the vicinity of the bond to be hydrolysed are all on only one side of the main chain.
Hide AbstractMcCleary, B. V., Matheson, N. K. & Small, D. B. (1976). Phytochemistry, 15(7), 1111-1117.
A series of galactomannans with varying degrees of galactose substitution have been extracted from the endosperms of legume seeds with water and alkali and the amount of substitution required for water solubility has been determined. Some were heterogeneous with respect to the degree of galactose substitution. The structural requirements for hydrolysis by plant β-mannanase have been studied using the relative rates and extents of hydrolysis of these galactomannans. A more detailed examination of the products of hydrolysis of carob galactomannan has been made. At least two contiguous anhydromannose units appear to be needed for scission. This is similar to the requirement for hydrolysis by microbial enzymes. Judas tree (Cercis siliquastrum) endosperm contained a polysaccharide with a unique composition for a legume seed reserve. Gel chromatography and electrophoresis on cellulose acetate indicated homogeneity. Hydrolysis with a mixture of β-mannanase and α-galactosidase gave a glucose-mannose disaccharide and acetolysis gave a galactose-mannose. These results, as well as the pattern of hydrolysis by β-mannanase were consistent with a galactoglucomannan structure.
Hide AbstractMcCleary, B. V. & Matheson, N. K. (1975). Phytochemistry, 14(5-6), 1187-1194.
Structural changes in galactomannan on germination of lucerne, carob, honey locust, guar and soybean seeds, as measured by viscosity, elution volumes on gel filtration and ultra-centrifugation were slight consistent with a rapid and complete hydrolysis of a molecule once hydrolysis of the mannan chain starts. β-Mannanase activity increased and then decreased, paralleling galactomannan depletion. Multiple forms of β-mannanase were isolated and these were located in the endosperm. β-Mannanase had limited ability to hydrolyse galactomannans with high galactose contents. Seeds containing these galactomannans had very active α-galactosidases. β-Mannosidases were present in both endosperm and cotyledon-embryo and could be separated chromatographically. The level of activity was just sufficient to account for mannose production from manno-oligosaccharides.
Hide AbstractMcCleary, B. V. & Matheson, N. K. (1974). Phytochemistry, 13(9), 1747-1757.
Germinating seeds of lucerne, guar, carob and soybean initially depleted raffinose series oligosaccharides and then galactomannan. This depletion was accompanied by a rapid increase and then a decrease in α-galactosidase levels. Lucerne and guar contained two α-galactosidase activities, carob three and soybean four. One of these in each plant, from its location in the endosperm, time of appearance and kinetic behaviour, appeared to be primarily involved in galactomannan hydrolysis. This enzyme in lucerne had MW of 23 000 and could not be separated from β-mannanase by (NH4)2SO4 fractionation, DEAE, CM or SE-cellulose chromatography or gel filtration, but only by polyacrylamide gel electrophoresis. In guar, carob and soybean, it could be separated by ion-exchange chromatography and gel filtration. In lucerne, carob and guar most of the total increase in activity was due to this enzyme. The other α-galactosidases had MWs of about 35 000 and could be separated from β-mannanase by dissection, ion exchange cellulose chromatography and gel filtration. They were located in the cotyledon-embryo and appeared to be primarily involved in galactosylsucrose oligosaccharide hydrolysis.
Hide Abstract